Reports: UNI355342-UNI3: Catalytic Hydrogenations Using First Row Transition Metals
William McNamara, College of William and Mary
Hydrogenation Results:
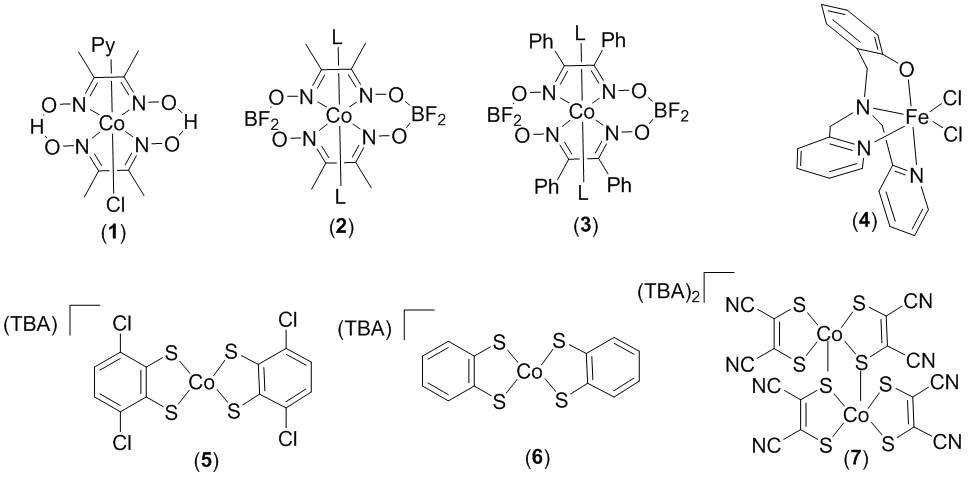
During our first year of funding, we have synthesized the complexes represented in Figure 1. These compounds represent a series of cobalt and iron hydrogen generation catalysts that operate at various redox potentials. With a diverse range of electrochemical potentials for hydride formation, this library of catalysts presents a very interesting group of complexes to examine for catalytic hydrogen transfer.
Figure 1. Cobalt and iron complexes as potential hydrogen transfer catalysts.
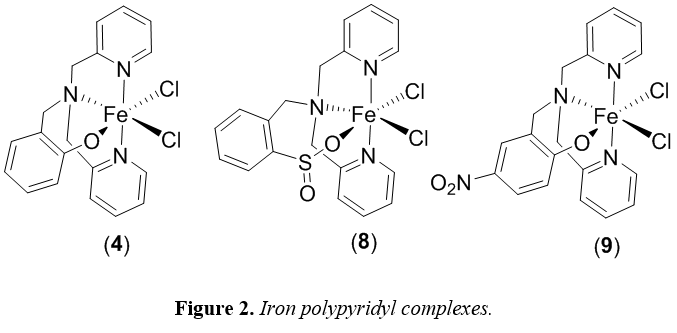
These complexes were synthesized by three undergraduate researchers and fully characterized using NMR, ESI-MS, and X-ray crystallography (for new compounds). When using isopropanol as the hydrogen source, all complexes in Figure 1 demonstrated modest activity (at best) for transfer hydrogenation of activated substrates. The substrates examined included methyl cinnamate, benzoquinone, acetophenone, and norbornene. Of these complexes, 4 showed the highest activity for the transfer hydrogenation of benzophenone (57% conversion, 200 turnovers). Although conversion was observed, these iron catalysts do not compare favorably to noble metal catalysts that have been previously reported. We then synthesized analogues of 4 (Figure 2) that had different electrochemical properties in an attempt to increase the activity of these complexes for transfer hydrogenation. Unfortunately, we found that complex 4 was the most active catalyst for all substrates tested. Ongoing research focuses on the incorporation of amine functional groups near the active site of these catalysts.
Proton Reduction Results:
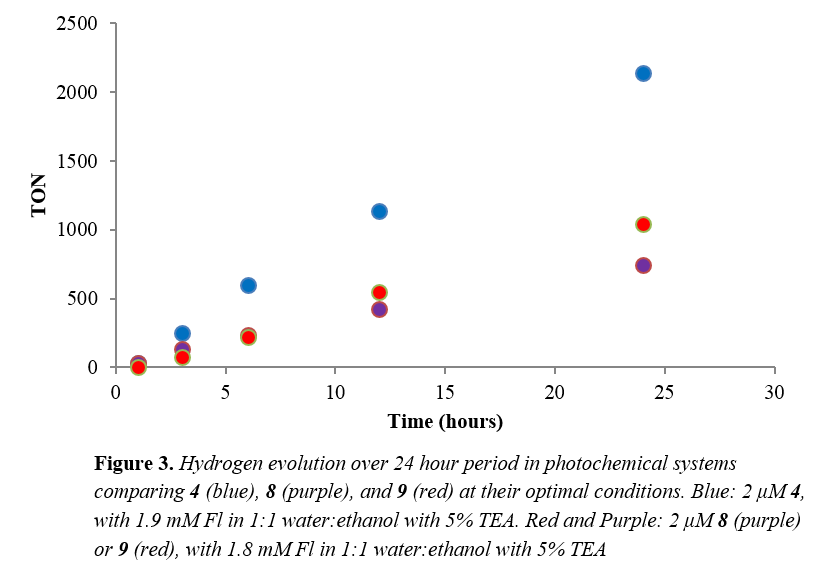
With complex 4, 8, and 9 already synthesized and fully characterized for use in transfer hydrogenation catalysis, we also tested their applicability as photocatalysts for proton reduction catalysis. We found that when paired with fluorescein as a chromophore and triethylamine as the sacrificial electron donor, these systems generate hydrogen for over 24 hours, achieving turnovers greater 2000 with respect to catalyst. This system is both highly active and stable for hydrogen generation using visible light.
Impact
This ACS-PRF grant provided the necessary funding to jumpstart our project on transfer hydrogenation chemistry using first-row metal complexes. It has allowed my group to purchase the materials needed to conduct this research. Furthermore, this grant has provided support for two undergraduate summer students (Mariah Frank and Joshua Tubb) to conduct research in this area during the summer of 2016. Another student (Teri Liu) was not funded directly by the grant, but worked during the summer of 2016 on this project. A poster on this research was presented by the PI at the Organometallics Gordon Research Conference during the summer of 2016. The PI will also give an oral presentation of this research at the South East Regional Meeting of the ACS (SERMACS) in October, 2016. Two undergraduate students will also present posters at SERMACS on this work in October, 2016. The research experience helped the summer students learn critical thinking skills as well as specific chemical techniques such as NMR, IR, UV-Vis, electrochemistry, and X-ray crystallography. Both Joshua Tubb and Teri Liu are currently applying to graduate schools to pursue a PhD in chemistry upon graduation in the spring. During the first year of funding, our work has been published in Polyhedron, Dalton Transactions, and Inorganic Chemistry.
Future Directions
Our group is currently synthesizing variations of our iron catalysts that contain amine functional groups that are in close proximity to the active site of the catalyst. Pendant amines have been found to increase the activity of certain ruthenium catalysts for transfer hydrogenation. Once synthesized, the pendant amine catalysts will be used for transfer hydrogenation catalysis of ketones and alkenes. H2 oxidation using these complexes will also be examined using electrochemical techniques.