Reports: ND351971-ND3: Metal-Free Activation of Nitrogen Oxides for Hydrocarbon Functionalization
Timothy H. Warren, Georgetown University
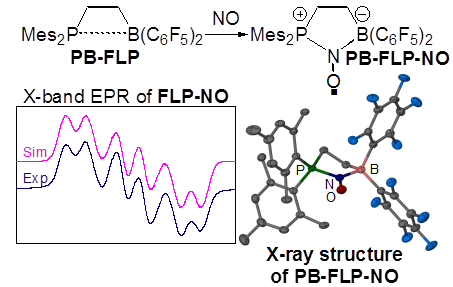
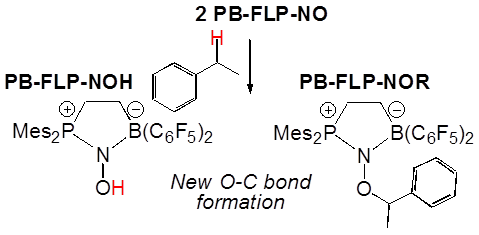
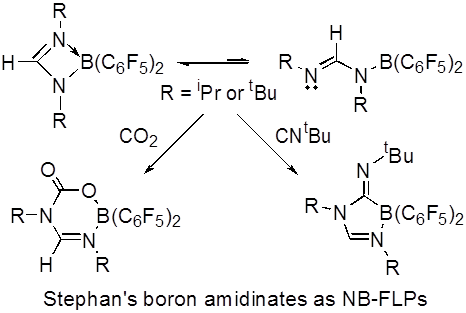
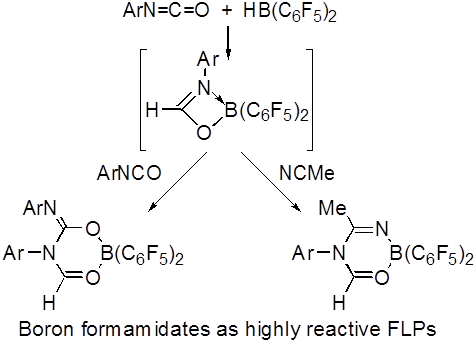
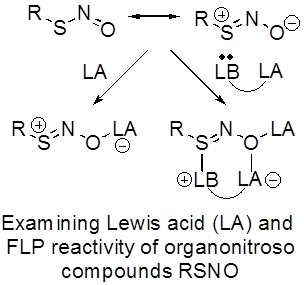
Timothy H. Warren, Georgetown University



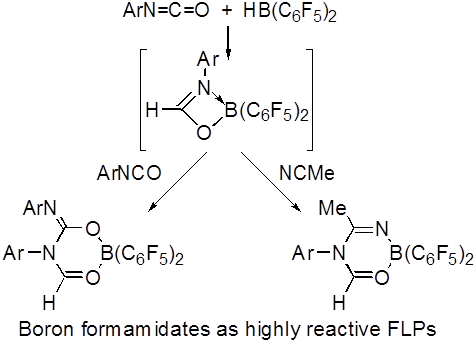

Reports in the ACS PRF Annual Report are published as submitted by the Principal Investigator.
Copyright © American Chemical Society