58th Annual Report on Research 2013 Under Sponsorship of the ACS Petroleum Research Fund
Reports: ND750941-ND7: Tetrazines for Hydrogen Storage
Douglas A. Loy, PhD, University of Arizona
Tetrazine Polymers for Hydrogen Storage
The goals of the project were to synthesize the bis(dimethylpyrazole)tetrazine monomer (1), prepare polymers from 1 based on nucleophilic displacement of the dimethylpyrazole groups, and begin to study the hydrogenation/dehydrogenation chemistry of the tetrazine ring system. The solubility of tetrazines in different polymers was also evaluated to explore the possibility of hydrogen storage in solid solutions. If successful in demonstrating reversible hydrogen in the materials, more studies of its utility for hydrogen storage were to be conducted. What we discovered was a rich field of chemistry and materials based on the highly colored tetrazines with numerous potential applications, including self-developing photoresists, new classes of thermosets with color indicators, and more.
Monomer synthesis
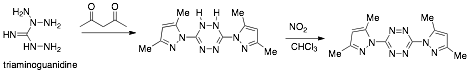
The monomer was synthesized by condensing triaminoguanidine with 2,4-penatandione to the dihydrotetrazine followed by oxidation to the desired tetrazine 1 (scheme 1). The dihydrotetrazine oxidized to 1 upon standing in air. Due to its extended conjugation, 1 was found to be a striking red hue (Figure 2), a characteristic typical for these heterocyclic molecules.
Scheme 1. Synthesis of tetrazine monomer 1
Tetrazine 1 and its derivatives were bright red or orange crystalline materials (Figure 1). The hydrogenated tetrazines were pale yellow (Figure 2).
Figure 1. 3,6-bis(3,5-dimethyl-1H-pyrazol-1-yl)-s-tetrazine (1)
Tetrazine hydrogenations
Samples of 1 and 3,6-diphenyl-1,2,4,5-tetrazine 2 were hydrogenated with one atmosphere of hydrogen in the presence of chloroplatinic acid. Upon addition of even two hydrogens to the tetrazine ring, the compounds decolorized to a pale white appearance similar to the dihydrotetrazine intermediate we isolated in the synthesis of 1 (Figure 2).
Figure 2. 3,6-bis(3,5-dimethylpyrazol-1-yl)-1,2-dihydro-1,2,4,5-tetrazine
The progress of the hydrogenation could be readily followed with in-situ infrared spectroscopy (ReactIR, and by the bleaching of the red solution to a pale yellow. With introduction of oxygen, the hydrogenated products quickly oxidized back to the respective tetrazines indicating that the hydrogen can be recovered.
Tetrazine solutions in polymers
It occurred to us during our investigations, that it may not be necessary to prepare polymers of tetrazines in order to effect solid state storage. If the tetrazines can be dissolved in polymers, particularly those with glass transition temperatures lower than room temperature, realtively simple hydrogen storage systems could be prepared more readily and potentially with greater quantitites of hydrogen. To this end, we examined the solubility of the tetrazines in molten polymers and in polymers already dissolved in solvents known to dissolve tetrazines 1 or 2. However, while some tetrazines 1 and 2 were soluble at elevated temperatures, the materials crystallized out with cooling to room temperture. Better results were obtained with polystyrene (Tg 100° C), polyethylene oxide (Tg -35°C, Tm 51 °C, Figure 3.), and polybutadiene (Tg -80°C). Polystyrene provided a glassy matrix for tetrazine 2 and Pd-C catalyst that showed not apparent decolorization upon exposure to hydrogen gas. Tetrazine 2 and Pd-C in polyethylene oxide, on the other hand, showed decolorization with exposure to hydrogen.
Figure 3: Hydrogenation of 2 in polyethylene oxide using chloroplatinic acid as the catalyst. The dark pink cylinder represents the control sample, and the light yellowish pink cylinder is a sample after hydrogenation.
However, even in the low glass transition polymer solutions, the rates of hydrogenation were generally too slow (hours) to provide effective hydrogen storage. Reoxidation of the reduced tetrazines back to their highly colored aromatic versions was shown to proceed quickly in solution, but appears to be very slow in solid state polymers. Our efforts to improve the rate of hydrogenation and dehydrogenation are focussing on solvated gels as the storage media rather than glassy polymers or elastomers.
Polymerization experiments
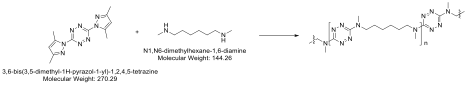
Polymerization of the tetrazines occurs by nucleophilic substitution of the dimethylpyrazole groups from the 3 and 6 positions on the aromatic ring. We discovered that like epoxy thermosets primary amines can react with two different tetrazine rings, to make network polymers. Model compound studies aided in this discovery. Polymerization of 1 with 1,2-diaminoethane or 1,6-diaminohexane resulted in insoluble, crosslinked tetrazine based polymers. We discovered that it was possible to copolymerize 1 with bis-amine monomers to afford linear soluble polymers (Scheme 2).
Scheme 2. Polymerization of tetrazine monomer 1 with diamine co-monomer
We also discovered that 1 will not polymerize with diol or dithiol comonomers, even with base catalysis. We did discover that 3,6-dichlorotetrazine 3, that is known to react with dithiols and diols, would copolymerize to form linear oligomers and polymers. The copolymer of 3 with ethane dithiol is of particular interest as we can oxidize the sulfide groups in the polymer backbone to the sulfones that are succeptible to photochemical degradation. We are presently evaluating the polymers UV stability and the potential utility of these materials as self-developing photoresists.
Diels-Alder Chemistry of Tetrazines.
Tetrazines hae been shown to react with dienophiles in [4 + 2] cycloaddition reactions with subsequent [4 +2] retro-Diels-Alder elimination of dinitrogen. We have studied the reactivity of tetrazines with different functional groups and the reactivity of the tetrazine as a diene relative to the pyridazine (diazabenzene). Theoretically tetrazines should be able to undergo two Diels-Alder cycloadditions and dinitrogen elimination reactions to form the nitrogen free aromatic product. This would allow tetrazines to be used as monomers in Diels-Alder polymerization reactions leading to polyarylenes. Model studies began late in the project and are continuing to date.
Conclusions
The project afforded a new family of polymeric materials based on tetrazine functional groups. This includes copolymers based on diamino- dithiol and dihydroxy functionalized comonomers. We have demonstrated hydrogenation and dehydrogenation of tetrazine functionalities in low glass transition temperature polymers, but have concluded that the rates in these systems are too slow to be useful for fuel storage. We have also shown that the polymers prepared from ethane dithiol can be oxidized to the polyemr with sulfones in the backbone, a potential new class of self-developing photoresists. Future hydrogen storage efforts will focus on improving hydrogen transport by incorporating the tetrazine functionalities into gels made from polymer and solvent. Diels-Alder polymerization chemistry of tetrazines will be used to prepare new polyarylenes and to crosslink new thermoset materials.
Copyright © 2014 American Chemical Society