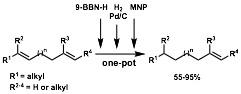58th Annual Report on Research 2013 Under Sponsorship of the ACS Petroleum Research Fund
Reports: UR150719-UR1: Regioselective Semihydrogenation of Dienes
Brian C. Goess, Furman University
Prior to our work on this project, there existed no general strategy for the direct, regioselective semihydrogenation of dienes that is selective for the more highly substituted alkene of a differentially substituted diene. Our project proposal was based on promising preliminary results indicating that a one-pot, reversible hydroboration strategy would be effective for simple diene substrates and had the following five objectives:
(1) Determining the Functional Group Tolerance of the Regioselective Semihydrogenation Methodology
(2) Determining the Substrate Scope of the Regioselective Semihydrogenation Methodology
(3) Expanding the Methodology to Include Chemoselective Hydrogenolysis
(4) Adapting the Methodology into a General Strategy for the Isomerization of Stilbenes
(5) Expanding the Synthetic Potential of Ring-Closing Enyne Metathesis (RCEM)
During the first grant year of this project, we achieved Objectives 1 and 4, which resulted in two publications. A summary of these accomplishments is provided in the preceding Narrative Report. During this first award period, I was delighted to be awarded tenure and a full-year sabbatical, which I spent away from campus at Northwestern University and Merck Pharmaceuticals. Accordingly, for reasons of safety and practicality, my undergraduate research lab was not in operation during the past academic year. Upon my return this past summer, I recruited new research students and resumed work on proposal objective 2.
Objective 2
9-BBN-H was selected as an alkene protective group for three principal reasons: (1) monohydroboration of differentially substituted terminal dienes is exceptionally regioselective for the terminal alkene when bulky dialkylboranes are used, leaving the more highly substituted alkene intact, (2) trialkylboranes are hydrogenated under heterogeneous catalysis only at elevated temperatures and pressures, and (3) trialkylboranes can be cleaved at elevated temperatures or in the presence of certain electrophiles, revealing the original alkene and producing an oxidized borane. This suggests a three-step hydroboration, hydrogenation, oxidation sequence to accomplish the desired regioselective transformation.
Since the first two steps of this sequence occur without production of stoichiometric byproduct, we envisioned a one-pot transformation involving sequential addition of 9-BBN-H, followed by hydrogen and catalytic Pd/C, and finally an electrophile. We evaluated a number of commercially available electrophiles and selected the powerful electrophile 2-methyl-2-nitrosopropane (MNP) because it oxidized the intermediate trialkylboranes quantitatively and within minutes to reveal the initial alkene. If ethanolamine is added following the final oxidation step, boron-ethanolamine complexes precipitate out of solution. Thus, purification frequently requires no more than filtration through a plug of silica gel to remove Pd/C and insoluble boronates, the only reaction byproduct. This one-pot procedure leads to the regioselective semihydrogenation of a variety of dienes.
Regioselectivity is high; generally only one product alkene regioisomer is observed by NMR. Synthetically useful yields are obtained for a variety of substrates including cyclic and acyclic terminal dienes, dienes with various substitution patterns, and strained internal dienes. Importantly, a conjugated terminal diene was semihydrogenated without constitutional isomerization of the remaining olefin, which is usually observed when such reductions are attempted with homogeneous and hetereogeneous catalysts. To assess the scope of this transformation, we have synthesized six new potential regioselective semihydrogenation substrates and will evaluate their reactivity in our semihydrognation reaction.
Impact of the Work
Two of the five papers that I published pre-tenure were directly funded by this grant, and I consider ACS-PRF funding to have been an essential component of both my successful tenure application and my successful application for a full-year, fully-funded sabbatical. So far, four undergraduates have been funded through this grant. One is now attending graduate school in organic chemistry at the University of Michigan, one is now in medical school at the University of Tennessee, one is a junior and plans to attend graduate school in organic chemistry, and one is a sophomore with currently undecided plans.
Copyright © 2014 American Chemical Society