58th Annual Report on Research 2013 Under Sponsorship of the ACS Petroleum Research Fund
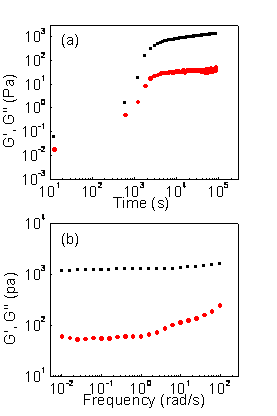
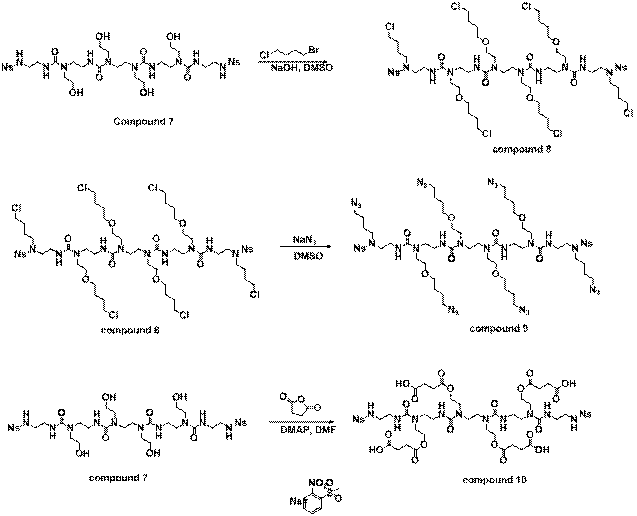
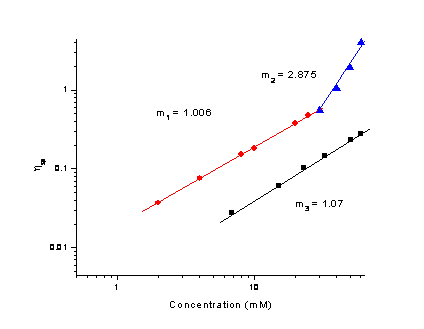
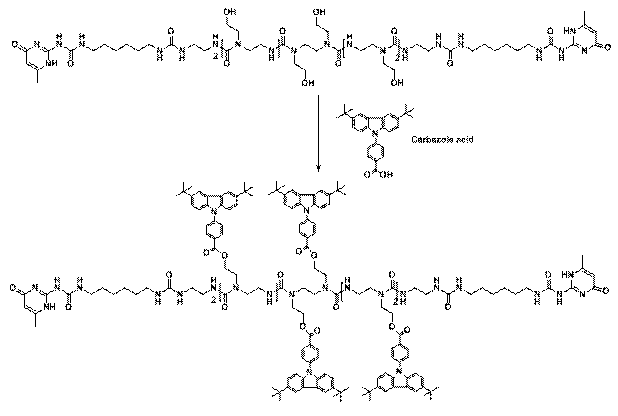
Reports: DNI751850-DNI7: N-Alkyl Urea Peptoid Oligomers as a New Type of Self-Assembling, Highly Versatile Soft Materials for Applications Involving Organogelators
Neil Ayres, PhD, University of Cincinnati

| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | ||
Copyright © 2014 American Chemical Society