58th Annual Report on Research 2013 Under Sponsorship of the ACS Petroleum Research Fund
Reports: UNI151259-UNI1: The Design, Synthesis, and Characterization of 'Smart' Donor-Acceptor Biaryls
Bartholomew James Dahl, PhD, University of Wisconsin (Eau Claire)
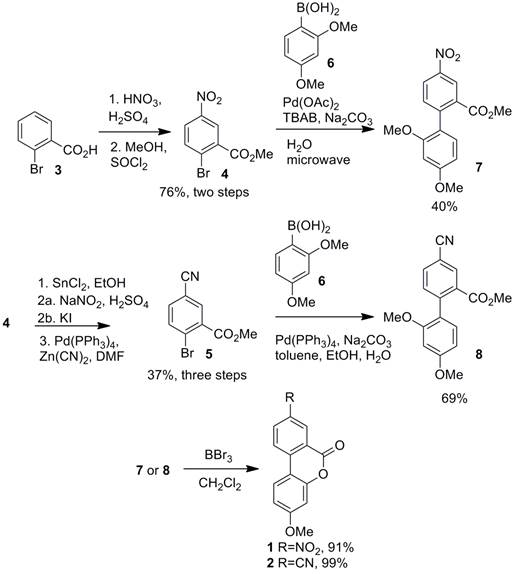
Research Area 1: Synthesis and Characterization of Donor-Acceptor Biaryl Lactone pH-Driven Molecular Switches
pH-driven biaryl lactone dihedral angle switches 1 and 2 have been synthesized following the steps shown below. Two undergraduate student researchers, Asia Riel and Erick Carlson (both supported internally and by the PRF), performed all of the synthetic work. Both students presented their work at spring 2012 ACS conference in San Diego (supported by PRF funds) and Asia again at the spring 2013 ACS conference in New Orleans (supported by PRF funds). Asia has gone on to graduate school at the University of Montana and Erick is now in his second year of graduate school for medicinal chemistry at the University of Minnesota.
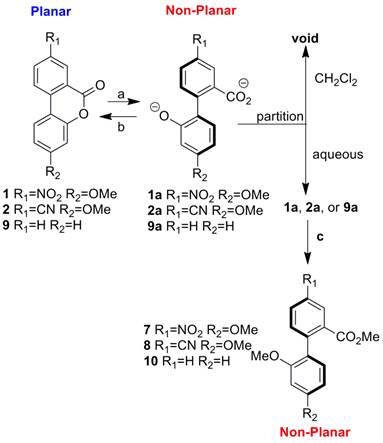
Both switches were determined to rapidly switch between the ring-opened (non-planar) and ring-closed (planar) states upon addition of acidic or basic stimuli. Compounds 1/2 ring open to 1a/2a in the presence of base and lactonize in the presence of acid. The figure below shows that process. Compounds 1a/2a were trapped as compounds 7/8 to confirm the identity of the ring-opened structures since they are water soluble and difficult to isolate.
These compounds are not only switches of dihedral angle but also of intramolecular charge transfer (ICT) between the donor and acceptor groups. Since the conjugation is attenuated in the ring opened state (1a and 2a), the charge transfer process is also attenuated. This process was readily probed by UV-Vis and fluorescence spectroscopy. The absorbance of compounds 1a and 2a are both greatly diminished relative to 1 and 2. We have also determined that the luminescence of 1 and 2 can be immediately diminished in the presence of base (switching to 1a and 2a) and then reformed after addition of acid. A paper has been published describing this project.
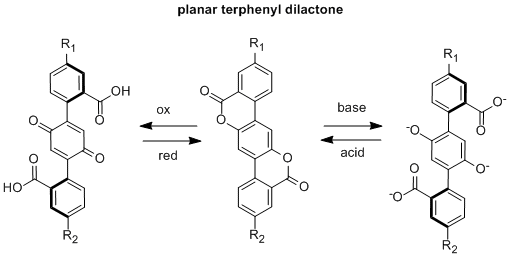
Research Area 2: Synthesis of Dual-Input Terphenyl Switches
We are currently working on the synthesis of terphenyl dilactones which should act as both redox and pH switches. This work began in summer 2012 and continued through the summer of 2013. Several student researchers, Asia Riel (supported by a McNair Scholarship in 2012), Zach Ahola (supported 100% by PRF in 2012), Justin Dressler (supported 100% by PRF in 2013), and Sarah Miller (supported 100% by PRF in 2013) have made major contributions to this project.
Our initial research goal was to synthesize unsymmetrical terphenyl dilactones in which R1 was a donor group and R2 was an acceptor group. These types of compounds should be pH and redox-driven switches of geometry and ICT. We found this synthetic route to be quite challenging despite attempting numerous synthetic strategies. However, symmetric terphenyl dilactones, where R1 = R2 should still act as molecular redox or pH switches, although they will not disrupt intramolecular charge transfer upon switching. We were successful in synthesizing a symmetric di-tert-butyl analog and found it does switch fluorescence via redox and pH. We are currently working toward synthesizing more of this compound so that we can begin more thorough testing of the switching process. Asia traveled to the spring 2013 ACS conference in New Orleans (supported by PRF funds) to present this research. A paper is currently is preparation for this project.
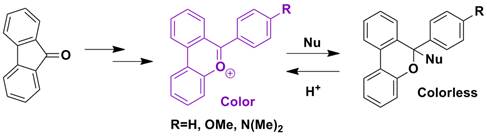
Research Area 3: Synthesis of Dibenzopyrylium Salts with pH Induced Optical Switching
Erin Prust and Erick Carlson both worked on this project in 2012 (both supported internally). Erin is now in Pharmacy School. Joel Patrow was supported 50% by the PRF in 2013 to work on this project. Joel traveled to the spring 2013 ACS conference in New Orleans (supported by PRF funds) to present this research. We have made several of these compounds and have shown they do switch visible absorbance at varying pH. Currently we are working on synthesizing more analogs to tune their optical properties. A paper has been published describing this project.
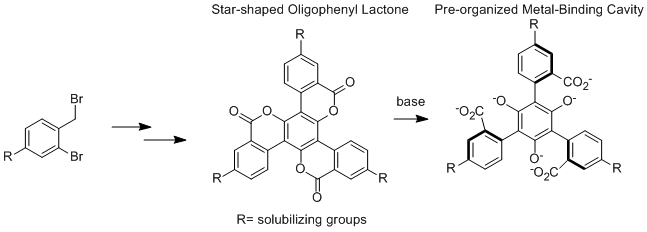
Research Area 4: Synthesis of Oligophenyl Lactones
One student, Nick Sortedahl, was supported (100% by PRF) on this project in 2012. Nick is now in graduate school for chemistry at Purdue University. Justin Dressler and Sarah Miller (both supported 100% by PRF in 2013) are currently making progress toward the synthesis of the compound below in which R = 3,5-di-tertbutylphenyl. We have found that simpler versions of these compounds have difficult solubility and are working toward resolving this issue. Nick traveled to the spring 2013 ACS conference in New Orleans (supported by PRF funds) to present this research.
The support from the ACS-PRF has been instrumental in getting these great research results. In my first three years at the University of Wisconsin-Eau Claire (UWEC) I have mentored ten students in research and many of them have been supported by the PRF. The students would have otherwise had a much smaller chance of getting involved in research. Now they have been introduced to the research process and also trained in practical advanced synthetic techniques and characterization. All of my summer research students get training in searching the literature, making presentations, and writing up professional experimental results. Opportunities were also available for two students to attend the spring national ACS conference in 2012 and three students at the spring meeting in 2013. Other students have presented posters on the UWEC campus for our Student Research Day. These opportunities have excited the students about doing research and most of my research students now plan to attend graduate school. Their performing research in my group will help better prepare them for what they will encounter in graduate school. Without the funding from the PRF, none of this would be possible. Additionally, the PRF has provided seed money for me to get my own research established at UWEC. Thus far, funding from the PRF has resulted in several conference presentations and two published articles in my two years of funding and other publications are currently being prepared. Additionally, I was invited to present the work of our group at the spring national ACS conference in 2013 in a symposium honoring Nancy Mills. The progress we made would not be possible without the PRF.
Copyright © 2014 American Chemical Society