58th Annual Report on Research 2013 Under Sponsorship of the ACS Petroleum Research Fund
Reports: UNI650746-UNI6: Rotational Spectroscopy Investigation of Interactions between Carbon Dioxide and N-containing Cyclic Compounds
Gordon G. Brown, PhD, Coker College
In this project, our main goal is to measure, assign, and analyze the rotational spectra of van der Waals complexes of carbon dioxide with four nitrogen-containing cyclic molecules: pyrimidine, pyridazine, pyrazine, and imidazole. The purpose of this study is twofold. First, we hope to better guide the next generation of materials developed for carbon capture and storage (CCS). Second, this study will provide an important test of previously published theoretical data.
Summary
In the second year of this project, we have successfully completed the following tasks:
i. Performed a second series of upgrades to the Coker College chirped-pulse Fourier transform microwave (CP-FTMW) spectrometer.
ii. Measured and analyzed two molecular compounds that have not been previously reported in the literature.
iii. Measured the spectra of several CO2-cyclic organic compounds, in pursuit of fulfilling the project’s main goals.
Spectrometer Upgrades
We have accomplished three upgrades to the Coker College CP-FTMW spectrometer in the previous year. First, we purchased and employed a 10 Watt solid-state power amplifier into our instrument. This amplifier replaces a 1 Watt amplifier, and will increase the signal-to-noise ratio on most systems of interest. The funds for this amplifier were provided by a recent grant from the National Science Foundation, which was an indirect result of the seed funding of the current PRF grant. Second, we have made changes to the geometry of the instrument which has resulted in higher signal to noise ratio on some systems. Finally, we have implemented a new timing system which allows us to record data ten times faster. Using this system, we are able to perform ten microwave measurements for every valve pulse; previously, we were performing a single microwave measurement for every valve pulse.
Experiments Performed
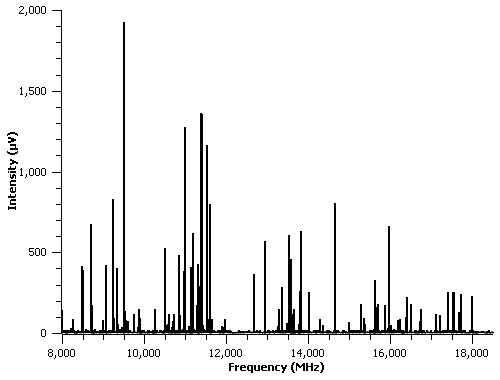
Students at Coker have measured and analyzed the spectra of the following compounds: 2-chloro-3-fluoropyridine and 2-chloro-6-fluoropyridine, diagrams of which are shown in Figure 1. These compounds tie into our laboratory’s interest in CO2-nitrogen containing organic ring complexes as they are nitrogen-containing organic rings. As an example, the pure rotational spectrum of 2-chloro-3-fluoropyridine is shown in Figure 2. This spectrum was measured after heating the sample to approximately 70 degrees Celsius and is the
result of 10,000 signal averages. Preliminary rotational and nuclear quadrupole coupling constants are shown in Table 1. Ab initio calculations have also been performed on these compounds; the results of the calculations are also shown in Table 1.
|
Table 1: Experimental Rotational Constants |
||||
Rotational Parameters |
2-chloro-3-fluoropyridine (35Cl) |
2-chloro-6-fluoropyridine (35Cl) |
||
|
B3LYP/ 6‑311++g(d,p) |
Experimental (uncertainty) |
B3LYP/ 6‑311++g(d,p) |
Experimental (uncertainty) |
A (MHz) |
2936.0358 |
2949.4203 (73) |
3412.8187 |
3420.8158 (16) |
B (MHz) |
1595.7478 |
1612.22822 (89) |
1245.8076 |
1258.4718 (14) |
C (MHz) |
1033.8474 |
1042.28067 (68) |
912.6544 |
919.91217 (61) |
3/2 Χaa N(MHz) |
0.74540 |
0.561 (92) |
2.73374 |
2.14 (11) |
¼ Χbb – Χcc N(MHz) |
-1.6796 |
-1.634 (33) |
-1.70307 |
-1.605 (18) |
3/2 Χaa Cl(MHz) |
-102.333 |
-107.421 (63) |
-86.401 |
-89.92 (12) |
¼ Χbb – Χcc Cl(MHz) |
2.8053 |
2.275 (22) |
-0.13151 |
-0.560 (21) |
Number of Assigned Lines |
N/A |
274 |
N/A |
138 |
OMC (MHz) |
N/A |
.01132 |
N/A |
.00939 |
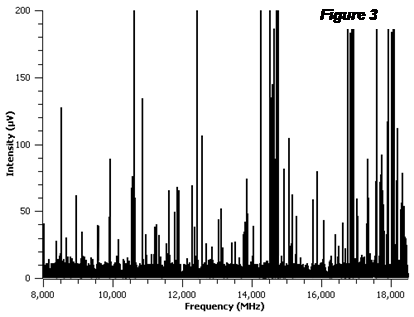
The main focus of the project is the study of carbon dioxide complexes with nitrogen-containing cyclic organic compounds. We have been able to measure spectra of several of the target complexes, but the analysis of these complexes has proved challenging. Therefore, we have identified new target systems not mentioned in the original proposal that will fulfill the overall goal of the project, but that should prove less ambitious than those proposed in the original proposal. We anticipate that these new systems will be easier to measure and analyze due to the presence of only a single nitrogen atom in the compound; thus, the spectra will not be diluted and complicated by multiple quadrupolar nuclei in the compound. These new systems include complexes of carbon dioxide with 2-fluoropyridine, 3-fluoropyridine, 3,5-difluoropyridine, and pyrrole. In Figure 3, the spectrum of the 2-fluoropyridine-CO2 complex is shown. This spectrum was generated after subtracting all of the lines from the pure 2-fluoropyridine spectrum, and it is the result of 50,000 averages. The analysis of the spectrum is ongoing.
We are continuing to work on the other complexes listed above as well as those listed in the original proposal. While the formal PRF project time period has finished, the overall idea of this project continues to be one of two main projects of the PI’s laboratory.
|
Figure 3
|
Copyright © 2014 American Chemical Society