Reports: UNI155237-UNI1: Supramolecular Catalyst Assembled through Aromatic Donor-Acceptor Interaction and Its Application in Hydrolytic Kinetic Resolution of Epoxides
Yu Liu, PhD, Northern Michigan University
Supramolecular catalysts refer to multicomponent assemblies that are stabilized by noncovalent interactions to achieve catalytic systems with high selectivity and high catalytic efficiency. The common noncovalent interactions used in the assembling supramolecular catalysts include metal-ligand complexation, hydrogen bonding, cation-anion attraction and ion-dipole interaction. The aromatic donor-acceptor interaction not only has a higher binding affinity and more precise control of spatial arrangement than the regular π- π interaction, but also tolerates a wide range of solvent from cyclohexane to water. In this project, we explored the assembly of supramolecular catalysts based on the aromatic donor-acceptor interaction, which has been rarely reported.
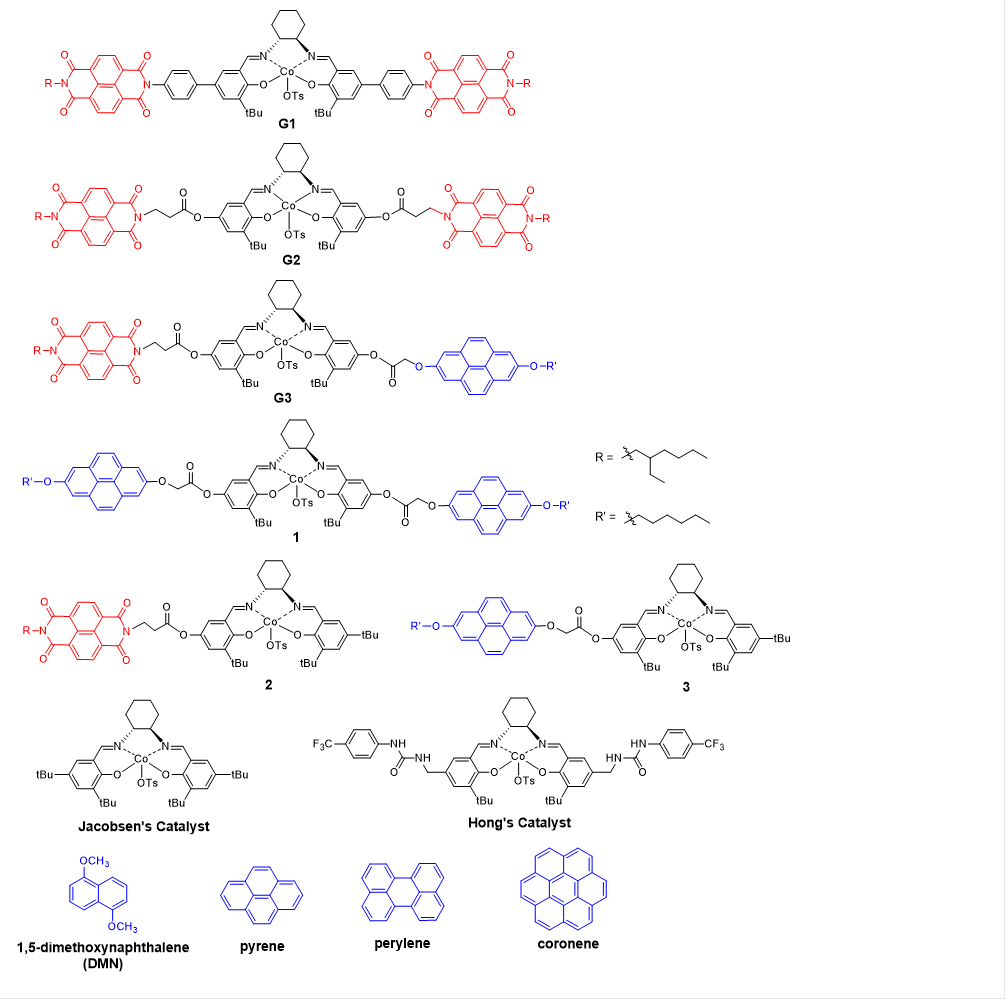
Figure 1. Co(III)-salen complexes, aromatic donor compounds and aromatic acceptor compound used or mentioned in this project.
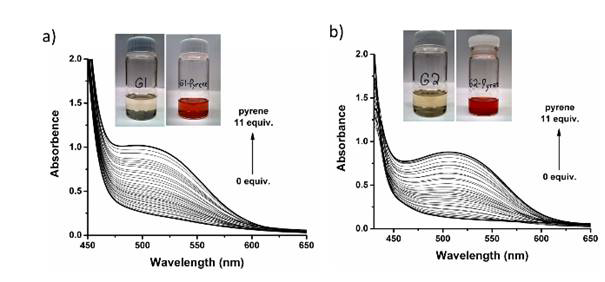
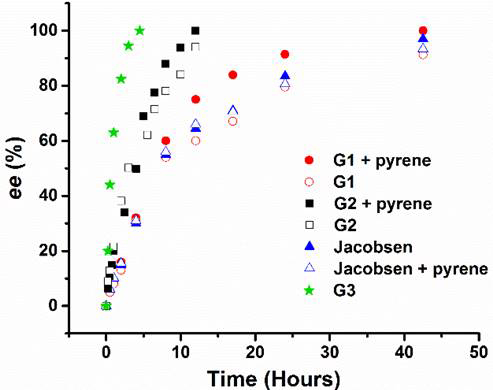
During the ACS-PRF grant funding period, three generations of Co(III)-salen complexes have been developed. Both Generation 1 (G1) and Generation 2 (G2) have a symmetric structure, in which the Co(III)-salen core is flanked by a pair of NDI (acceptor) as the recognition units (Figure 1). In G1 and G2, NDI connects to the central Co(III)-salen core via a rigid phenyl linker and a flexible and non-conjugated alkyl ester linker, respectively. It was expected that the introduction of electron-rich aromatic compounds (donor) enhanced cooperativity of the two Co(III)-salen complexes, and consequently accelerated the rate of HKR of epoxides. G1 and G2 were tested in HKR of epichlorohydrin under a variety of reaction conditions, such as different solvents and different donor compounds (pyrene, 1,5-dimethoxynaphthlene, perylene and coronene). Under the optimized reaction condition, it took 12 hours for G2 and 42.5 hours for G1 to complete the HKR of epichlorohydrin. The much higher catalytic activity of G2 was contributed to its flexible alkyl ester linker, which may allow the Co-salen cores to reach the preferred conformation for a cooperative pathway. For both G1 and G2, the catalyst systems with a donor compound showed higher catalytic efficiency than those without one, suggesting that the aromatic donor-acceptor interaction helps to promote the assembly of dinuclear Co-salen catalyst. The binding properties were investigated by the UV-vis titration of the corresponding Zn(II)-salen complexes of G1 and G2. The evolution of broad absorption bands was detected with maximum at 505 nm for G1-Zn complex and 510 nm for G2-Zn complex, upon addition of pyrene, characteristic of the absorption of the aromatic donor-acceptor interaction (Figure 2). The 1H NMR spectroscopy titration experiments showed the upfield shift of NDI protons, upon addition of pyrene, providing additional evidence of the aromatic donor-acceptor interaction.
Figure 2. (a) The UV-vis titration curves of G1-Zn with pyrene. Insert: the photographs of G1d solution (left) and the titration ending G1-Zn / pyrene solution (right). (b) The UV-vis titration curves of G2-Zn with pyrene. Insert: the photographs of G2-Zn solution (left) and the titration ending G2-Zn / pyrene solution (right).
To further improve the binding affinity, Generation 3 (G3) Co(III)-salen complex was designed and synthesized, which contains both the electron-rich pyrene and the electron-poor NDI, so the self-assembly of a supramolecular catalyst can be achieved. Under the optimized reaction condition, the HKR of epichlorohydrin by G3 was completed in 3.5 hours, which is more than ten times shorter than the reaction time by Jacobsen’s Catalyst (42 hours) and is also much faster than 14 hours by hydrogen bonding based Hong’s catalyst. A series of control experiments were performed. Under the same reaction condition, bis-NDI functionalized G2, bis-pyrene functionalized 1, mono-NDI functionalized 2 and mono-pyrene functionalized 3 need 7 to 12 hours to complete the reaction. However, both the 1:1 mixture of G2+1 and the 1:1 mixture of 2+3 completed the reaction in 4 hours, indicating the dinuclear assembly through the aromatic donor-acceptor interaction. These results clearly demonstrated the aromatic donor-acceptor interaction facilitated the dinuclear complex assemblies for both homo-complex (G3) and hetero-complexes (G2+1 and 2+3), and played an important role in their high HKR activity.
Figure 3. Kinetic plots of enantiomeric excess (ee) of HKR of epichlorohydrin by G1, G2 and G3.
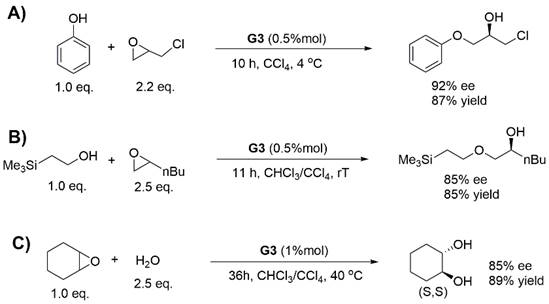
We also carried out the substrate scope tests of G3. G3 also showed outstanding catalytic activity and enantioselectivity in the HKR of the structurally diverse terminal epoxides (Table 1). These results demonstrated the excellent functional group tolerance of the aromatic donor-acceptor interaction. We further explored the reaction scope of G3 with asymmetric ring-opening of epoxides with nucleophiles other than water (Figure 4A and 4B) and asymmetric ring-opening of a non-terminal epoxide, cyclohexane epoxide (Figure 4C). The results demonstrated again the high catalytic efficiency and versatility of G3.
Table 2. HKR tests of G3 with various terminal epoxides.
R | Loading (% mol) | Solvent | Time (h) | ee (%) | Conv. (%) |
n-butyl | 0.05 | CCl4 | 6 | > 99 | 52 |
Ph | 0.5 | CHCl3 | 17 | 97.5 | 49 |
CH2Cl | 0.05 | CCl4 | 3.5 | > 99 | 53 |
ethyl | 0.05 | CHCl3/CCl4 (1:1) | 4 | > 99 | 54 |
COOCH3 | 0.25 | CHCl3 | 24 | 96 | 50 |
CH2OAllyl | 0.05 | CCl4 | 5 | > 99 | 51 |
Figure 4. Asymmetric ring-opening reactions by G3.
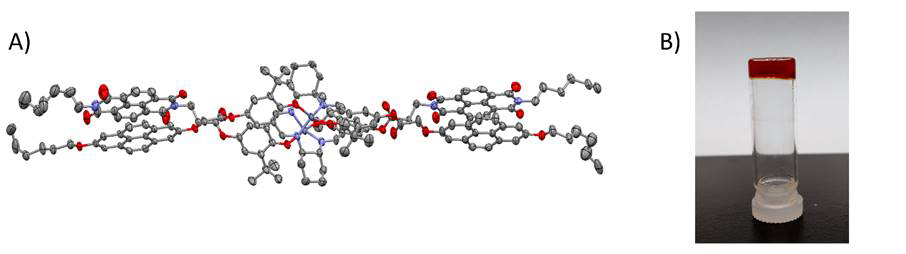
1H NMR dilution experiment was performed for G3 ligand. Protons in both NDI and pyrene moieties showed downfield shifts when the G3 concentration was decreased, which confirmed the aromatic donor-acceptor interaction. In the end, we also attempted to obtain the crystal structure of G3 assembly. Eventually, the G3’-Zn complex crystal grown from CHCl3/DMF was resolved (Figure 5A). The X-ray structure clearly showed the face-centered binding between NDI and pyrene. In the attempt to grow G3’-Zn crystal in CHCl3/cyclohexane, we obtained a red gel (Figure 5B). The aromatic donor-acceptor interaction could be the main noncovalent force in the gel formation.
Figure 5. (A) X-ray structure of G3’-Zn dimer complexes. (B) The photograph of G3’-Zn gel in CHCl3/cyclohexane.
During this funding period, nine NMU undergraduate students worked on this project. Four of them got the stipends funded by this PRF. Four posters on this work have been presented (2016 and 2017 ACS Upper Peninsular Local Meetings; 2016 ACS Central Regional Meeting; 2017 ACS Great Lake Regional Meeting). One manuscript has been submitted and another manuscript is under preparation.