Reports: UR356549-UR3: Development and Catalytic Evaluation of Palladium Complexes Bearing Modified Amine Bis(phenolate) Ligands
Bradley M. Wile, Ohio Northern University
Overview/Aims
The aims of this project are to identify structural modifications to the amine bis(phenolate) (ABP) ligand framework that may increase catalytic activity of related late transition metal catalysts. In the first year, we have prepared a number of ligand scaffolds and investigated the structure and bonding present in corresponding palladium(II) complexes.
Progress in Year One
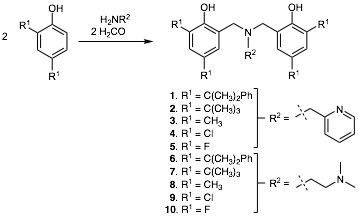
In the first year of this grant, our group prepared a number of ABP ligands with varied substitution patterns (see Figure 1), and their corresponding palladium complexes. Some ligands were prepared for the first time by our group, while other have been previously reported. Optimization of synthetic procedures has allowed for the preparation of multi-gram quantities of analytically pure ligands in good to high yield in most cases. Several other ligand variations have been determined to possess physical properties rendering them not amenable to isolation or further study.
Figure 1. Preparation of amine bis(phenolate) ligands under study by our group.
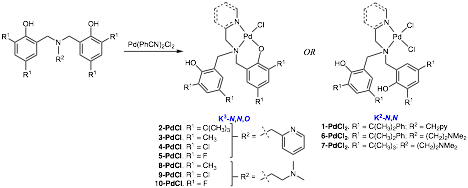
Combining equal molar quantities of [Pd(PhCN)2Cl2] and the appropriate diprotic ligand generated the target palladium(II) complexes with either bidentate (K2-N,N) or tridentate (K3-N,N,O) coordination modes (see Figure 2). Complexes were characterized using a combination of FT-IR, NMR (1H, 13C, 19F), melting point, elemental analysis, and in some cases single crystal X-ray diffraction experiments. Single crystal diffraction data collection, solution, and refinement for seven palladium complexes was conducted with the X-ray Diffraction Laboratory at Purdue University. Characterization of these complexes is complete with the exception of four compounds awaiting satisfactory elemental analysis. Spectroscopic characterization of these compounds is consistent with the depiction in Figure 2
Figure 2. K2 and K3 amine bis(phenolate) palladium(II) complexes.
Several key complexes have been modelled using density functional theory (B3LYP/LANL2DZ), and the resulting optimized geometries have been compared to those observed in the solid state by X-ray diffraction experiments. Solution studies to probe the pH dependent interconversion of K2 and K3 ABP complexes have been conducted and monitored by NMR. In addition, a survey of the reactivity of small molecules with 10-PdCl has been initiated, using 19F NMR to track speciation. We expect these studies will facilitate conclusions about the speciation of (ABP)Pd complexes throughout the catalytic cycle.
Student Impact and Dissemination
Four undergraduate students participated in this research project during the 2016/17 academic year using supplies furnished by this grant and/or Ohio Northern University. Two of these students have now graduated, and are enrolled in chemistry graduate programs at the University of Minnesota Twin Cities and Indiana University Bloomington, respectively. In the summer of 2017, two ONU students were primarily supported by this grant, one of whom had conducted research with me during the 2016/17 academic year. As a result of their involvement, these students gained experience in organic synthesis, purification, metallation, and spectroscopic characterization of small molecules. Funding two summer research students from this grant (year one) has greatly bolstered the summer research environment in the Donald. J. Bettinger Department of Chemistry and Biochemistry at ONU. Our department strategic planning calls for increased student involvement in mentored summer research activities, and we hope to leverage externally-funded summer positions to increase support from ONU donors. The summer research students have commented that they found the summer rewarding and enjoyable, particularly being able to commit an entire day to a research task instead of a few hours over the course of the week, as would happen during the academic year.
Progress on this project was documented in weekly or end-of-semester reports, and disseminated via presentations at two professional meetings. The PI contributed an oral presentation at the 253rd National Meeting of the American Chemical Society in San Francisco CA, and student co-authors presented posters describing this project at the 253rd National Meeting of the American Chemical Society and the 49th Midwest Theoretical Chemistry Conference in East Lansing MI. A manuscript describing our findings is in progress, and will include significant authorship from undergraduate students supported by PRF.