Reports: DNI755075-DNI7: The Living Anionic Polymerization of Functionalized Aziridines
Paul A. Rupar, PhD, University of Alabama
Background: Poly(ethylenimine) (PEI) is valuable for use in non-viral gene transfection,[1] CO2 capture,[2] and the formation of anti-microbial surfaces,[3] among many other applications.[4] Despite the resemblance of PEI to poly(ethylene oxide) (PEO), the polymerization of aziridine greatly differs from that of ethylene oxide. Whereas ethylene oxide polymerizes anionically to form linear PEO, aziridine exclusively undergoes cationic ring-opening polymerization (CROP) to produce hyperbranched PEI (bPEI).
Linear PEI (lPEI) can be attained via the CROP of N-substituted aziridines, however control via these procedures has not been demonstrated.[5] The standard route to lPEI is through the CROP of oxazolines to form poly(2-oxazoline)s (POx), followed by post-polymerization hydrolysis of the POx.[4a, 6] The POx route achieves some control over molecular weight and polydispersity, but chain transfer reactions during the polymerization and difficulties in the hydrolysis to lPEI remain problematic.
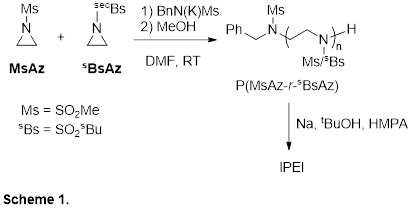
Research Progress: Our work has focused on developing the anionic ring-opening polymerization (AROP) of N-sulfonyl aziridines as a new route to linear PEI. A challenge encountered by us and others is that poly(N-sulfonylaziridines) suffer from poor solubility, thus making their synthesis problematic. We have overcome this difficulty by realizing that poly(N-sulfonylaziridine) random copolymers have greatly improved solubility compared to the homopolymers. Using a 1:1 ratio of two different sulfonyl aziridines (MsAz and sBsAz, see Scheme 1), the resulting random copolymer poly(MsAz-r-sBsAz) has greatly improved solubility and can be characterized and processed.
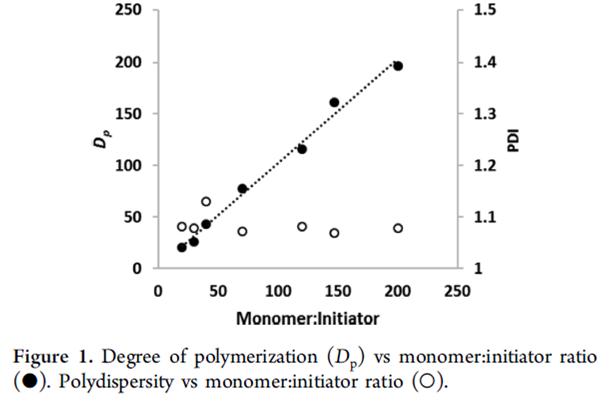
Since the copolymerization is a living AROP, we can target specific molecular weights by simply varying the ratio of the monomers to the anionic initiator (Figure 1). The polymers formed via this route have low molecular weight distributions, with dispersities typically less than 1.1. The polymerization is also amenable to the synthesis of block copolymers through sequential polymerizations.
On disadvantage to using sulfonylaziridines in the synthesis of linear PEI is that the removal of the sulfonyl groups requires harsh conditions. Over the past year, we have been examining alternative protecting groups that can activate aziridines for AROP, but are easier to remove post-polymerization. One of the new protecting groups is nitrophenyl sulfonyl (nosyl). A distinct advantage of the nosyl group is that it can be removed under mild conditions, specifically using a thiol in the presence of a base.[7]
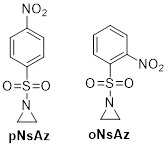
Nitrophenyl sulfonyl has two common isomers, para-nitro substituted and ortho-nitro substituted. We have synthesized two new aziridine monomers, pNsAz and oNsAz (Scheme 2), based on these two isomers. Both pNsAz and oNsAz undergo AROP to form the polysulfonylaziridines poly(pNsAz) and poly(oNsAz). Based on previous experience with polysulfonylaziridines, it is not surprising that we found poly(pNsAz) to have very poor solubility. Interestingly, poly(oNsAz) is soluble in polar solvents, a fact that we attribute to the reduced symmetry of the oNsAz group, which we hypothesize disrupts interchain packing. As is characteristic of nosyl protected amines, we were able to remove the nosyl groups under mild conditions from poly(oNsAz).
We are now applying our knowledge gained from the polymerization of sulfonylaziridines to the related azetidines. The polymerization of azetidines mirrors that of aziridines in that polymerizations are typically cationic and produce hyperbranched polymers. We recently synthesized N-(methanesulfonyl)azetidine and are currently exploring its polymerization chemistry (Scheme 2).
Scheme 2.
Two graduate students in my group are supported by this grant: Canisius Pmbarushimana and Louis Reisman. Canisius is expected to graduate with a PhD in 2018 and Louis with a PhD in 2019. Both students are progressing well in their studies and their theses will be based on the research supported by this grant. The grant has also provided funds to travel to conferences to present this research.
[1] (a) A. Falco, P. Encinas, S. Carbajosa, A. Cuesta, E. Chaves-Pozo, C. Tafalla, A. Estepa, J. M. Coll, Fish Shellfish Immunol. 2009, 26, 559-566; (b) J. H. Jeong, S. H. Song, D. W. Lim, H. Lee, T. G. Park, J. Control. Release 2001, 73, 391-399; (c) A. Akinc, M. Thomas, A. M. Klibanov, R. Langer, J. Gene. Med. 2005, 7, 657-663; (d) Z. Kadlecova, S. Nallet, D. L. Hacker, L. Baldi, H.-A. Klok, F. M. Wurm, Macromol. Biosci. 2012, 12, 628-636; (e) M. A. Mintzer, E. E. Simanek, Chem. Rev. 2008, 109, 259-302.
[2] (a) J. H. Drese, S. Choi, R. P. Lively, W. J. Koros, D. J. Fauth, M. L. Gray, C. W. Jones, Adv. Funct. Mater. 2009, 19, 3821-3832; (b) S. Satyapal, T. Filburn, J. Trela, J. Strange, Energy Fuels 2001, 15, 250-255.
[3] (a) S. Kobayashi, Prog. Polym. Sci. 1990, 15, 751-823; (b) V. P. Dhende, S. Samanta, D. M. Jones, I. R. Hardin, J. Locklin, ACS Appl. Mater. Interfaces 2011, 3, 2830-2837.
[4] (a) M. Jäger, S. Schubert, S. Ochrimenko, D. Fischer, U. S. Schubert, Chem. Soc. Rev. 2012, 41, 4755-4767; (b) G. F. Zou, J. Zhao, H. M. Luo, T. M. McCleskey, A. K. Burrell, Q. X. Jia, Chem. Soc. Rev. 2013, 42, 439-449; (c) T. M. McCleskey, P. Shi, E. Bauer, M. J. Highland, J. A. Eastman, Z. X. Bi, P. H. Fuoss, P. M. Baldo, W. Ren, B. L. Scott, A. K. Burrell, Q. X. Jia, Chem. Soc. Rev. 2014, 43, 2141-2146.
[5] (a) P. K. Bossaer, E. J. Goethals, Die Makromol. Chem. 1977, 178, 2983-2988; (b) J. R. Demember, L. D. Taylor, J. Polym. Sci., Part A: Polym. Chem. 1979, 17, 1089-1101; (c) K. F. Weyts, E. J. Goethals, Polym. Bull. 1988, 19, 13-19.
[6] L. Tauhardt, K. Kempe, K. Knop, E. Altuntaş, M. Jäger, S. Schubert, D. Fischer, U. S. Schubert, Macromol. Chem. Phys. 2011, 212, 1918-1924.
[7] T. Kan, T. Fukuyama, Chem. Commun. 2004, 353-359.