Reports: ND251785-ND2: Elucidation of Reactions Mediated by Sulfidic Carbonate and Clay Depositions: The Search for New Organic Reactions Mediated by Natural Materials
Salvatore D. Lepore, PhD, Florida Atlantic University
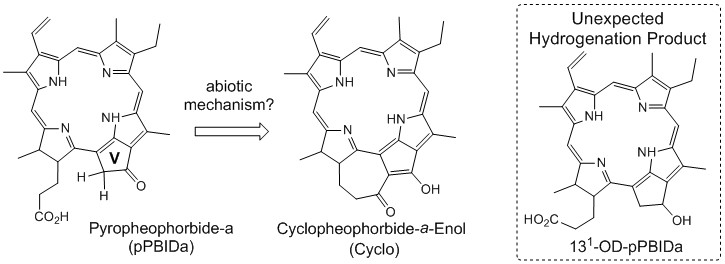
Salvatore D. Lepore, PhD, Florida Atlantic University

Copyright © American Chemical Society