57th Annual Report on Research 2012 Under Sponsorship of the ACS Petroleum Research Fund
Reports: UNI1050237-UNI10: Controlling Morphology and Electronic Properties of Two-Dimensional Organometallic Conjugated Polymers via Orthogonal Polymerization Methods
Katsu Ogawa, PhD, California State University (Northridge)
Development of new and efficient materials for organic solar cells has become one of the most important areas of chemistry. The performance of photovoltaic devices highly depends on efficiencies of absorption, charge separation, and charge migration. Our research goal is to improve charge migration efficiency by constructing 2D conjugated polymer matrix. In our approach, phosphole type ligands are incorporated in place of typical trialkyl phosphine ligands for platinum acetylide polymers. Unlike its nitrogen analogue (pyrrole), the lone pair on phosphorus atom is not completely associated with the pi system of the phosphole ring. As a result, phosphole exhibit non-planar structure with ligating capability through the lone pair. On the other hand, phospholes can be polymerized via oxidation just as pyrroles and thiophenes. The introduction of phosphole ligands in Pt-acetylide polymers results in possibility for cross linking a conjugated polymer (Pt-acetylide) with another conjugated polymer (Polyphosphole). Such cross linking should lead to increased conjugation, hence the improved charge migration for enhanced efficiency of the photovoltaic devices.
First Year Accomplishment
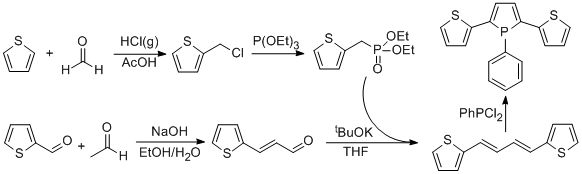
A new synthetic route to 1-phenyl-2,5-bis(2-thienyl)phosphole was developed using 1,4-bis(2-thienyl)butadiene as a precursor. Treatment of thiophene with paraformaldehyde in the presence of HCl forms 2-(chloromethyl)thiophene in moderate yield. This compound is reacted with triethyl phosphite to give diethyl(2-thienylmethyl)phosphonate in 70-80% yield. Aldol condensation of 2-thiophenecarboxaldehyde with acetaldehyde gives 3-(2-thienyl)-2-propenal in 20-30% yield. Wittig type reaction with this aldehyde and diethyl(2-thienylmethyl)phosphonate gives the precursor 1,4-bis(2-thienyl)butadiene in 60-80% yield. A few attempts were made to synthesize 1-phenyl-2,5-bis(2-thienyl)phosphole.
Second Year Accomplishment
The effort was focused on optimization of the above synthetic scheme. Initial attempts to synthesize 1-phenyl-2,5-bis(2-thienyl)phosphole ligand resulted in very low yields (2~3%). Major byproduct was insoluble polymeric material that is most likely due to polymerization of the precursor diene and/or the desired product. Another major byproduct seem to be an oligomer, which is inseparable from the desired product by conventional separation technique such as column chromatography. The effect of the temperature on byproduct formation has been investigated extensively. Above 160 °C oligomeric byproduct formation is favored. Below 150 °C, oligomer formation is negligible, however, the conversion of the starting material to the product is very slow that the reaction requires 3 days to complete. Although oligomer formation can be suppressed by lowering the temperature, a significant amount of polymeric material is still present. So far the optimal condition gave 14% yield of pure desired product.
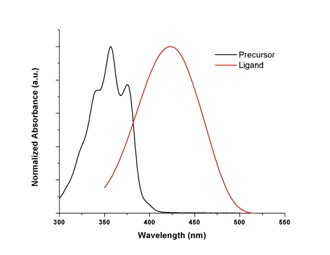
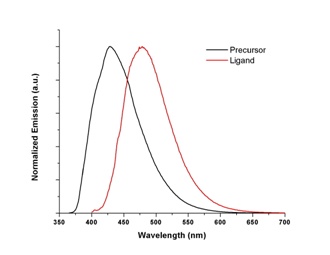
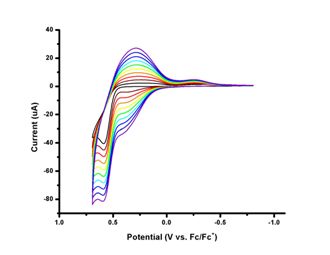
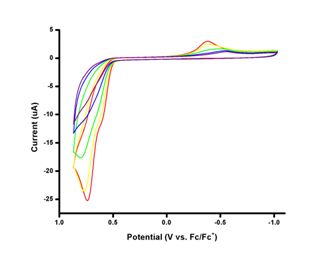
Photophysical and electrochemical characterizations have been performed on both the precursor 1,4-bis(2-thienyl)butadiene and the ligand 1-phenyl-2,5-bis(2-thienyl)phosphole. As seen below, bathochromic shifts of absorption (~60 nm) and emission (~70 nm) maxima are observed upon annulation of butadiene moiety by introduction of phosphorus atom. Extended pi system results in lower HOMO-LUMO energy gap. Interestingly, oxidation potential of the ligand (+741 mV) is higher than that of the precursor (+588 mV). Upon cycling of the potentials, the precursor molecule can be successfully polymerized as expected for thiophene containing molecules. The ligand, however, the oxidation occurs at the phosphorus atom of phosphole ring instead of the sulfur atom of thiophene ring. Thus, it leads to lower currents for later scans unlike that of polymerization in which higher currents are observed for later scans. Similar phenomena have been observed for other phosphole containing thiophene oligomers. Since coordination to a metal should prevent oxidation at phosphorus atom, synthesis of a platinum complex with this ligand is underway.
The Impact of the ACS PRF UNI Grant on the PI and Students
The UNI grant provided essential financial support for PI's research activities. The continuation of the research would have been impossible without UNI grant after the start up funding from the institution was exhausted for purchasing spectroscopic analytical instrumentations. Total of eight undergraduate students were actively involved in the project during the semesters. Four of these students are still active in this project.
Plans for the Third Year
The priority is given to preparation of platinum metal complex. The ligand 1-phenyl-2,5-bis(2-thienyl)phosphole will be treated with K2PtCl4 to obtain a platinum complex bearing chloride ligands and phosphole based ligands. Then photophysical and electrochemical characterization of the complex will be performed. This complex will be chemically polymerized via Hagihara coupling using chloride ligands on the platinum in the presence of diethynylbenzene. The resulting Pt-acetylide polymer will be spin-coated onto an ITO electrode and attempts will be made to cross link the polymer via electrochemical oxidation. The prepared 2D polymer matrix will be characterized by spectroscopic and electrochemical techniques. The preparation and characterization of platinum complex should be accomplished in FAll 2012. Characterization of polymeric materials are to be done in Spring and Summer 2013.










