57th Annual Report on Research 2012 Under Sponsorship of the ACS Petroleum Research Fund
Reports: DNI750918-DNI7: The Effect of a Polyelectrolyte on the Oppositely Charged Colloidal Suspension
Chongli Yuan, PhD, Purdue University
Stabilization of colloidal suspension has important applications in industry. Polymer additives, polyelectrolytes in particular, have been used as an efficient reagent in modulating the stability of colloidal particles. A polyelectrolyte can differently bind to a colloidal surface and lead to a distinctive adsorption and oligomerization pattern in a colloidal suspension. The goal of this project is to elucidate the roles of a polyelectrolyte in mediating the critical interaction that leads to the stabilization of a colloidal suspension.
To achieve this goal we have two specific aims: 1) reveal the micro-structure of a poly-electrolyte and colloidal particle mixture and 2) build a computer simulation model to predict the effects of a polyelectrolyte on the oppositely charged colloidal suspension.
Summary of results
1. Monitor the end-to-end distance of a polyelectrolyte under different colloidal suspension conditions
We primarily focused on a model system consisting of protein colloidal particles with positive surface charges and DNA (polyelectrolyte) with negative surface charges.
Förster resonance energy transfer (FRET) was adopted to monitor the end-to-end distance of a polyelectrolyte in colloidal suspensions with different surface charge properties and ionic strengths. The results were summarized in Fig.1. First, the size of a polyelectrolyte was reduced by >50% in a colloidal suspension as compared with that in a buffer of an identical ionic strength (~15nm). These results suggest that the polyelectrolyte were adsorbed to the oppositely charged colloidal surface and can wrap around the particle surface. Second, as the effective surface charge of the colloid was reduced by ~10%, the end-to-end distance of the polyelectrolyte was increased by ~20%. The attractive interactions between colloids and polyelectrolytes, therefore, facilitate the two ends of a polyelectrolyte to come closer to each other. Instead of assuming a static conformation, the polyelectrolyte adopts a conformation with flexibility primarily in the end region. Third, we evaluated the effect of buffer ionic strength on the polyelectrolyte conformation. With increasing salt concentrations, we observed a rapid increase in the DNA end-to-end distance. The findings of the FRET experiments reveal the conformation of a polyelectrolyte in a colloidal suspension. The polyelectrolyte is found to form close contacts with the colloidal surface and exhibit reduced backbone rigidity. In addition, a polyelectrolyte assumes a dynamic conformation with end fragments constantly breathing away from a colloidal surface at low salt concentrations.
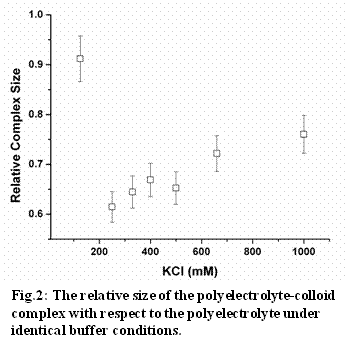
2. Quantify the size of colloid-polyelectrolyte complexes
To examine the stability of a colloidal suspension, we utilized fluorescence correlation spectroscopy (FCS) to monitor the size of colloid-polyelectrolyte complex. Specifically, FCS measures the hydrodynamic size of a complex by quantifying the translational diffusivity. Fig.2 summarized the effect of ionic strength on the colloidal-polyelectrolyte complex. The addition of polyelectrolytes to the colloidal suspension leads to the adsorption of polyelectrolytes to colloidal surfaces. In spite of the increase in mass, the colloid-polyelectrolyte complex was observed to migrate at a higher diffusivity than that of the polyelectrolyte alone. Decreasing ionic strength enhances the polyeletrolyte-colloidal particle interactions. As a result, further compaction of the complex was observed at lower salt concentrations. However, as the ionic strength decreases further, an increase in complex size was observed and this increase is likely due to the formation of colloidal aggregates bridged by polyelectrolytes.
3. Establish a photon counting histogram approach to explore the nucleation of phase transition
A colloidal suspension tends to exhibit lower stability primarily due to the formation of aggregates. To monitor the aggregation process at molecular level, we established an experimental approach based on the photon counting histogram (PCH) to follow the formation of colloidal aggregates at early nucleation stage. This approach quantifies the molecular brightness of each complex in solution and can be used to identify the relative population of multimers existing at different ionic strengths and interaction forces. The measured brightness of each diffusing species allows us to accurately calculate the amount of colloidal particles that are involved in the nucleation process of aggregate formation. The existence of a dimeric colloid (tagged with a fluorescence probe) can be distinctively detected as a molecular species with a molecular brightness quantified 1.5±0.3 times that of a monomer.
Overall Impact
1. We have successfully revealed the microscopic conformational features of a polyelectrolyte in colloidal suspension. Our findings elucidate the molecular mechanism that accounts for the interactions between a polyelectrolyte and an oppositely charged colloidal particle. These results have been presented in two national conferences and have led to two manuscripts that are currently under review.
2. We established a fluorescence based platform to examine the nucleation and growth of colloidal particle aggregates under different polyelectrolyte concentrations and ionic strengths. This platform equips us with a powerful tool to explore the phase-transition like behavior of colloidal suspension and provides us sufficient molecular details to construct a Brownian Dynamic simulation model to account for the transition behavior of colloidal suspensions.
3. This project provides an excellent opportunity to train graduate students with engineering background. One graduate student was supported by this grant. She has received the essential training on colloidal science and fluorescence microscopy. She designed and performed the experiments along with the PI and is currently working on the simulation codes for accomplishing the second aim of this project.
4. This project provides the PI with the essential resources to establish her lab and recruits her first graduate student. The findings of this project have led to the production of significant preliminary data for the PI to seek grant supports from other federal agencies.