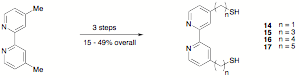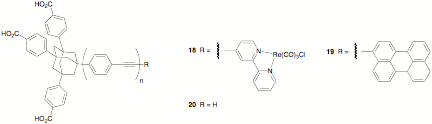AmericanChemicalSociety.com
Reports: B6 45966-B6: Dependence of Rates of Interfacial Electron Transfer on Anchoring Group STructure and Dye MLCT State Energy
Debra L. Mohler, James Madison University
Because there has been no systematic study of the effects on ET rate of the identity/properties of the anchoring group or of the electronic coupling between the adsorbate and the semiconductor (through matching of the energy levels of the dye to that of the semiconductor conduction band), the proposed research entailed the systematic design, synthesis, and photoinduced interfacial ET studies of a series of chlorotricarbonylrhenium bipyridine complexes substituted on the bipyridine ligand with a variety of anchoring groups (complexes 1 – 9), substituents that modulate the energy levels of the ligand p-molecular orbitals (molecules 10 – 13), or that vary the distance between the dye and the nanoparticle surface (compounds 14 – 18).
In the
first year of the grant, we prepared 1, 3, and 9; and in collaboration with Professors Tianquan Lian
and Keiji Morokuma at Emory University, we published femtosecond IR and
computational studies on their interfacial ET behavior. In addition, we also
completed the synthesis of Re complex 8 and of the bipyridine ligands in compounds 4 and 5.
In year 2 of the grant, we attempted to study the ET behavior of 8; and femtosecond IR studies with 8 have been hindered by difficulties getting this
complex to bind to any nanoparticles. The bipyridine ligands in 4 and 5
were found to decompose under all conditions with which we have attempted to
convert them to their chlororhenium tricarbonyl complexes. Because of these difficulties, we
abandoned the preparation of complexes 4 – 7; and instead synthesized 14 – 17 and their chlororhenium tricarbonyl complexes. We published this
synthetic work, and studies on the ET behavior of the chlororhenium tricarbonyl
complexes of these ligands are underway for comparison to our previously
published results on analogs with carboxylate anchoring groups. However,
because the tethers in both the thiol complexes 14 – 17 and their carboxylate analogs are flexible, leading to ambiguity in the
distance between the rhenium complex and the nanoparticle surface, we have
successfully prepared complexes of the type 18 (n = 1, 2, and 3)
and we have begun to synthesize similar molecules (19, n = 1, 2, and 3). These molecules have a tripod
anchoring group to position the phenylene-ethynylene bridge perpendicular to
the nanoparticle surface, allowing for an accurate determination of distance
between the surface and the dye. At this point, we have prepared the precursor 20,1 as well
as 1-bromoperylene;2 and to obtain 19 with n = 1, 2, or 3, we need only couple 20 to 1-bromoperylene and hydrolyze the ethyl esters
protecting the carboxylic acid groups.
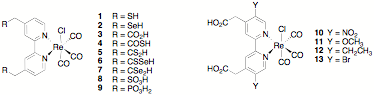
1 Galoppini, E.; Guo, W.; Zhang, W.; Hoertz, P. G.; Qu, P.; Meyer, G. J. J. Am. Chem. Soc. 2002, 124, 7801–7811. Guo, W.; Galoppini, E.; Rydja, G.; Pardi, G. Tetrahedron Lett. 2000, 41, 7419–7421.
2 Maeda, H.; Nanai, Y.; Mizuno, K.; Chiba, J.; Takeshima, S.; Inouye, M. J. Org. Chem. 2007, 72, 8990–8993.
Copyright © American Chemical Society