Reports: UR452071-UR4: Hydrogen-Bonding Control of Solvatochromism and Non-Radiative Decay in the Fluorescence of PRODAN Derivatives
Christopher J. Abelt, PhD, College of William and Mary


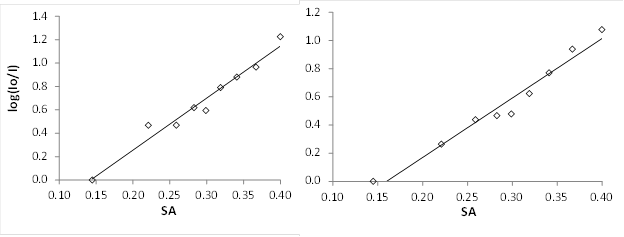
Christopher J. Abelt, PhD, College of William and Mary



Copyright © American Chemical Society