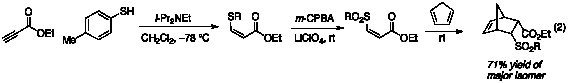AmericanChemicalSociety.com
Reports: GB1 48322-GB1: Synthesis of Chiral Carboxylic Acid Derivatives via Three-Component Coupling Reactions with Ynoate Electrophiles
Chiles Wade Downey, University of Richmond
Introduction
Our goal is to control the one-pot addition of two disparate nucleophiles to a single ynoate acceptor in order to yield a chiral carboxylic acid derivative (eq 1). Thus, simple ynoate electrophiles may be converted in one reaction vessel to densely functionalized, stereochemically complex cyclic or polycyclic systems. Successful development of this method represents a valuable advance for a number of reasons: 1) Ynoate esters are readily available, via either carboxylation of acetylene or oxidation of propargyl alcohols. 2) Three-component coupling reactions are highly efficient. 3) These reactions rapidly construct complex synthons for target-oriented synthesis.
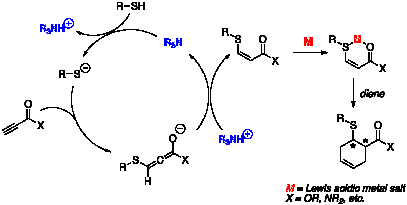
A proposed
mechanism for a representative heteroconjugate addition-Diels–Alder
process is outlined in Figure 1.
First, a trialkylamine base (R3N) deprotonates a thiol
nucleophile, generating a thiolate anion and a trialkylammonium cation. Conjugate addition by the thiolate
provides an allenolate intermediate, and the allenolate is protonated by the
trialkylammonium salt to yield an enoate ester. The enoate ester then acts as a conjugate acceptor; in the
example below, it is activated by a Lewis acid (M) and undergoes
Diels–Alder cycloaddition.
Our ultimate goal is to control the formation of the new stereocenters
by the use of a chiral auxiliary or a chiral metal catalyst. Figure 1. Mechanism for Three-Component
Coupling Reaction of Ynoates Current Results We plan a
three-pronged approach toward the development of this three-component coupling
reaction: 1) establishment of the amine-catalyzed conjugate addition of various
nucleophiles to ynoates, 2) use of the conjugate addition products as
dienophiles in Diels–Alder cycloadditions, and 3) development of an
asymmetric variant. To date, the
first goal has been accomplished and great progress toward the second goal has
been achieved. Goal 3 awaits optimization
of the current racemic process (vide infra). 1. Amine-Catalyzed Conjugate Addition to Ynoates Our
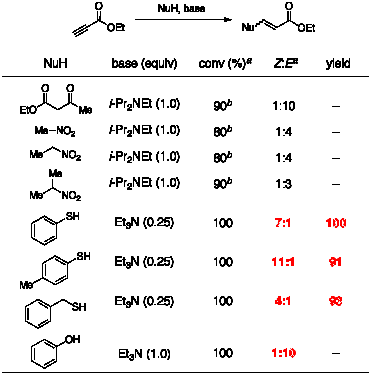
investigation of amine-catalyzed conjugate addition to ethyl propiolate shows
promising results, as described in our preliminary proposal (Table 1). Our early focus has been on thiol
nucleophiles, which require only catalytic amounts of amine and afford
excellent yields with high Z:E ratios. Very recently, we have expanded the
scope of this reaction to include phenol as a nucleophile, producing a vinylogous ester product. Table 1. Preliminary Results for Amine-Mediated Addition to Ethyl
Propiolate aDetermined by 1H NMR spectroscopy of the unpurified reaction
mixture bAfter workup with aqueous acid 2.
Thioether Oxidations Although
production of these thioethers occurs reliably, the thioether adducts
themselves are not active in Diels–Alder reactions under thermal or Lewis
acidic conditions. We directed our
efforts toward a one-pot two-step thioconjugate addition-oxidation sequence, to
produce a sulfoxide or sulfone product under conditions that would also be
amenable to Diels–Alder cycloaddition. We found meta-chloroperbenzoic acid (m-CPBA) to be a convenient oxidant for our system because of its
compatibility with our solvent of choice, CH2Cl2. After verification that the purified thioether could be
oxidized with m-CPBA, conditions for
the optimal production of either the sulfoxide or the sulfone were developed
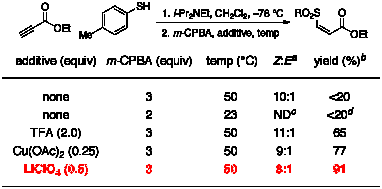
(Table 2). We found that 2 equiv m-CPBA was
sufficient to achieve oxidation to the sulfoxide, and 3 equiv produced the
sulfone. Despite high conversion
(>90%), however, isolated yields for these processes were consistently lower
than 20%. Close inspection of the 1H
NMR spectrum of the unpurified reaction mixture revealed a large number of
unidentified byproducts. Table 2. One-Pot Thioconjugate Addition-Oxidation Reaction aDetermined by 1H NMR
spectroscopy of the unpurified reaction mixture bIsolated yield after chromatography cND = Not Determined dyield of sulfoxide We
speculated that byproduct formation was caused by residual amine from the
thioconjugate addition step.
Indeed, oxidation of the purified thioether proceeded in high yield with
no byproduct formation. The
negative effect of the amine, however, could be mitigated by the addition of
acidic additives. Trifluoroacetic acid (TFA), Cu(OAc)2, and LiClO4 were all effective
additives, but we chose LiClO4 for further studies because of its
compatibility with the Diels–Alder reaction we envisioned as the third
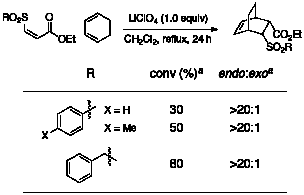
step of our one-pot process. 3. Diels–Alder Cycloadditions The
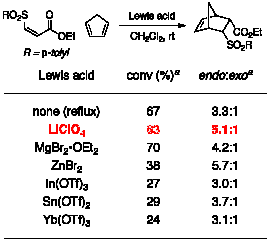
Diels–Alder portion of our reaction was first optimized using the purified
sulfone oxidation product. A
survey of Lewis acid catalysts at various temperatures showed LiClO4
to be the optimal catalyst for cycloaddition by cyclopentadiene, providing the
product in high conversion and high endo:exo
ratio (Table 3). Fortunately,
LiClO4 is also compatible with the reaction conditions for the
thioconjugate addition and oxidation reactions. Moreover, it catalyzes Diels–Alder reactions of sulfoxides, viable alternative dienophiles for our one-pot
process. Table 3. Cycloaddition of Cyclopentadiene to Z Sulfone aDetermined by 1H NMR spectroscopy of the unpurified reaction
mixture Very
recently, we expanded the diene scope to include cyclohexadiene. Again, LiClO4 was the
optimal catalyst (Table 4).
Surprisingly, no other catalysts proved capable of mediating
cycloadditions by cyclohexadiene.
Furthermore, only CH2Cl2 appears to be a viable
solvent at this date, for reasons that remain unclear. Nonetheless, the diastereoselectivity
for these reactions is remarkable: only the endo isomer can be detected by 1H
NMR spectroscopy. Table 4. Cycloaddition of Cyclohexadiene to Z Sulfone aDetermined by 1H NMR spectroscopy of the unpurified reaction
mixture After
discovering that LiClO4 positively affects the oxidation reaction as
well as the Diels–Alder cycloaddition, we modified our procedure for our
one-pot three-step thioconjugate addition-oxidation-Diels–Alder sequence. By adding the LiClO4
immediately prior to the oxidation
step, then continuing under our normal conditions, we were able to achieve a
71% yield of the major isomer for the cyclopentadiene reaction (eq 2). This yield is a substantial increase
over the 15% yield observed when no additive is present during the oxidation
step, and represents a promising precedent for expansion to other substrates. Conclusion We
have established an effective, high-yielding method for our model system, the
three-component coupling of ethyl propiolate, toluenethiol, and cyclopentadiene. We are now aggressively moving forward
with the expansion of the reaction scope to include other thiols and dienes,
and we have begun to investigate a one-pot vinylogous ester
formation-epoxidation process that also derives from ynoate esters. Our other long-term goals include the
development of asymmetric versions of our reaction.
Copyright © American Chemical Society