www.acsprf.org
Reports: DNI1048882-DNI10: Layered Rare-Earth Antimony and Bismuth Suboxides as Novel Thermoelectric Materials
Yurij Mozharivskyj , McMaster University
Objective:
The goal of the ACS-PRF funded project was to prepare novel thermoelectric materials via chemical fusion of semimetallic RESb/REBi and insulating RE2O3 binaries (RE is a rare-earth element). The idea was to structurally combine the RESb/REBi and RE2O3 phases with different properties in order to obtain "electron-crystal phonon-glass" materials with improved thermoelectric performance (Figure 1).
Figure 1. Formation of (RESb)n(RE2O3) from RESb, an "electron crystal", and RE2O3, a "phonon glass".
Results:
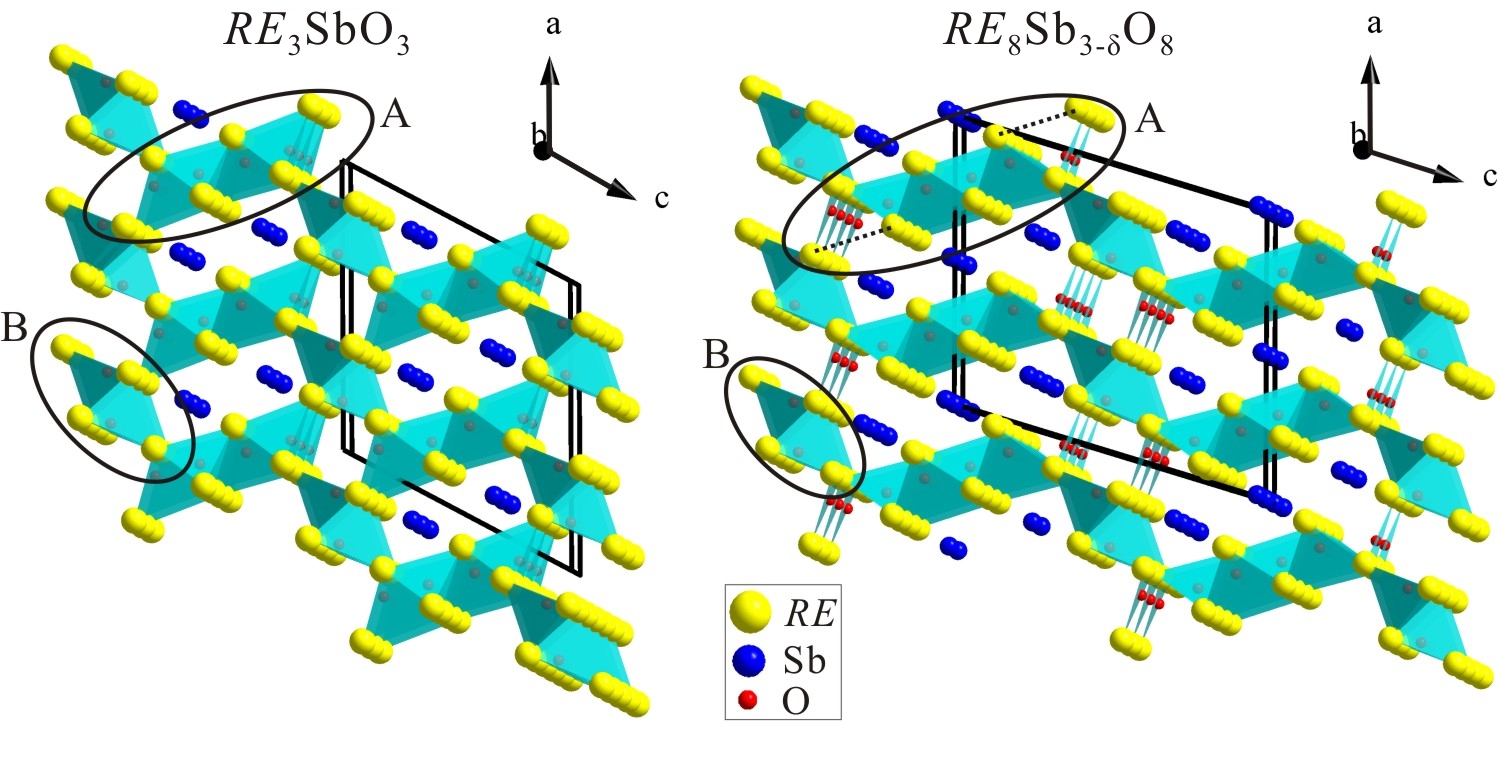
1) RE3SbO3 and RE8Sb3-d O8 antimonide oxides
In the search of high-temperature thermoelectric materials, two families of novel narrow band-gap semiconducting suboxides with the RE3SbO3 and RE8Sb3-d O8 compositions (RE = La, Sm, Gd, Ho) have been discovered (Figure 2). Their synthesis was motivated by attempts to open a band gap in the semimetallic RESb binaries through a chemical fusion of RESb and corresponding insulating RE2O3. Temperatures of 1350oC or higher are required to obtain these phases. Both RE3SbO3 and RE8Sb3-d O8 adopt new monoclinic structures with the C2/m space group and feature similar REO frameworks composed of "RE4O" tetrahedral units. In both structures, the Sb atoms occupy the empty channels within the REO sublattice. High-purity bulk Sm and Ho samples were prepared and subjected to electrical resistivity measurements. Both the RE3SbO3 and RE8Sb3-d O8 (RE = Sm, Ho) phases exhibit a semiconductor-type electric behavior. While a small band gap in RE3SbO3 results from the separation of the valence and conduction bands, a band gap in RE8Sb3-d O8 appears to result from the Anderson localization of electrons.
Discovery of RE3SbO3 and RE8Sb3-d O8 opens a new chapter in the chemistry of rare-earth suboxides, which was barely explored until now.
Figure 2. Structure of RE3SbO3 and RE8Sb3-d
O3
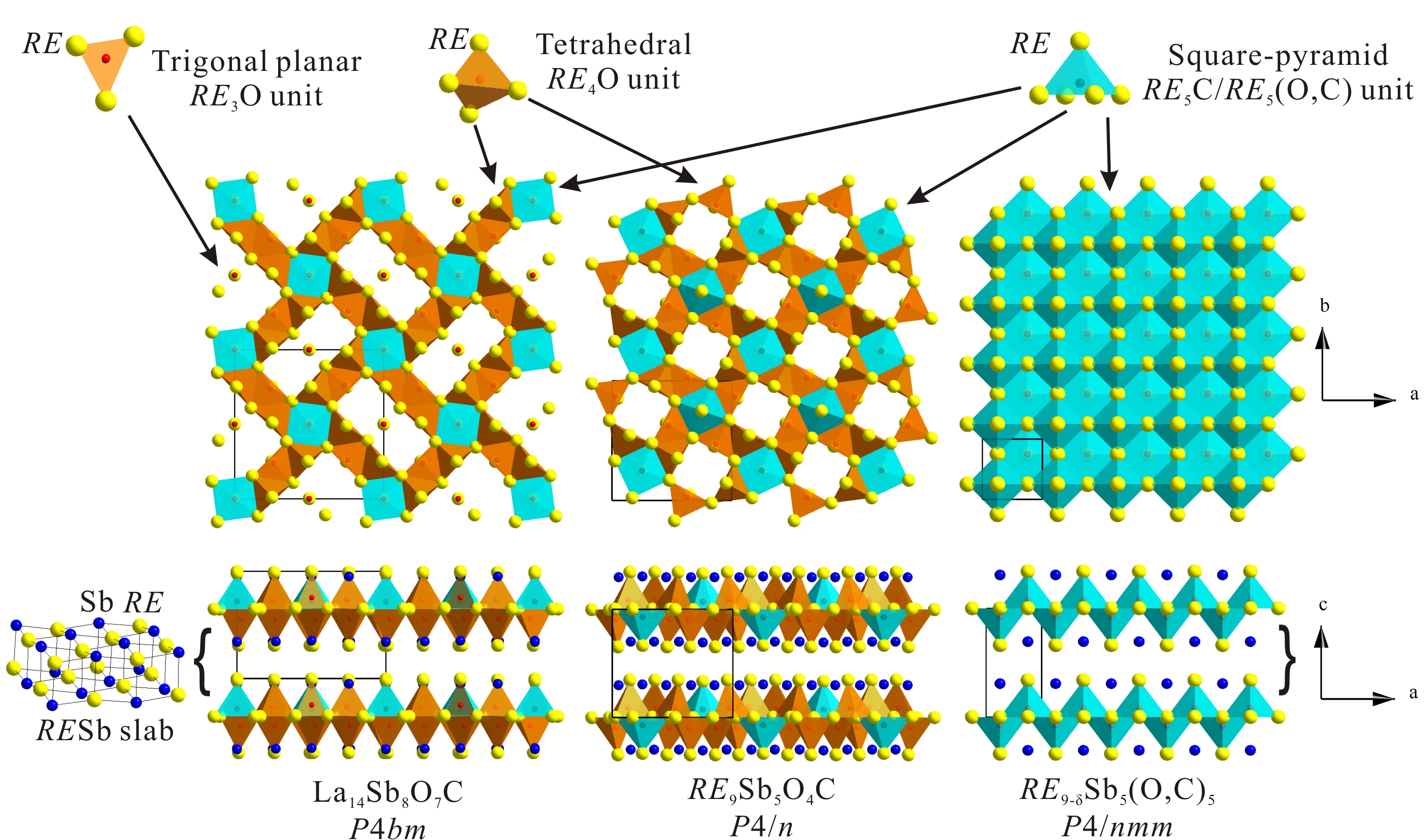
2) RE-Sb-O-C Natural Superlattice Phases (RE = La, Ho)
A family of the rare-earth antimonide oxycarbides have been prepared and structurally characterized. These superlattice phases are constructed from the NaCl-type RESb slabs sandwiched between the RE-O-C layers. Depending on the carbon content and synthetic conditions, three different RE-Sb-O-C structures can be obtained as shown in Figure 3. At lower temperatures, RE9-dSb5(O,C)5 phases with the P4/nmm symmetry are obtained for RE = La, Ho. A O/C mixture, with the O:C ratio of larger than 4:1, is randomly distributed within the RE-O-C layers. Also, the RE atoms are highly disordered within the oxide layer. At higher temperatures above the melting point of the samples, the RE9Sb5O4C phases with the P4/n symmetry are produced. The RE-O-C layers in RE9Sb5O4 are fully ordered: the RE sites are well defined and the oxygen and carbon atoms occupy the tetrahedral and square-pyramidal voids, respectively. Also at high temperatures, a new ordered La14Sb8O7C structure with the P4bm symmetry was discovered. The La14Sb8O7C phase is structurally similar to RE9Sb5O4C and features ordered arrangements of RE and O/C atoms in the La-O-C layer. The RE9-dSb5(O,C)5, RE9Sb5O4C and La14Sb8O7C phases appear to be charge balanced, and their compositions and structures are controlled by the O/C ratio. Parallel preparative experiments revealed the importance of carbon in the formation of these layered phases. In addition, it has been established that the purity of the rare-earth metals influences the compositions and structures of the products.
The layered nature of these phases suggests that other structures may be formed, e.g. with REBi slabs instead of RESb ones or thicker RESb slabs. Currently, the research efforts are conducted to test these ideas.
Figure 3. Crystal structures of La14Sb8O7C, RE9Sb5O4C, RE9-dSb5(O,C)5 and their building blocks.
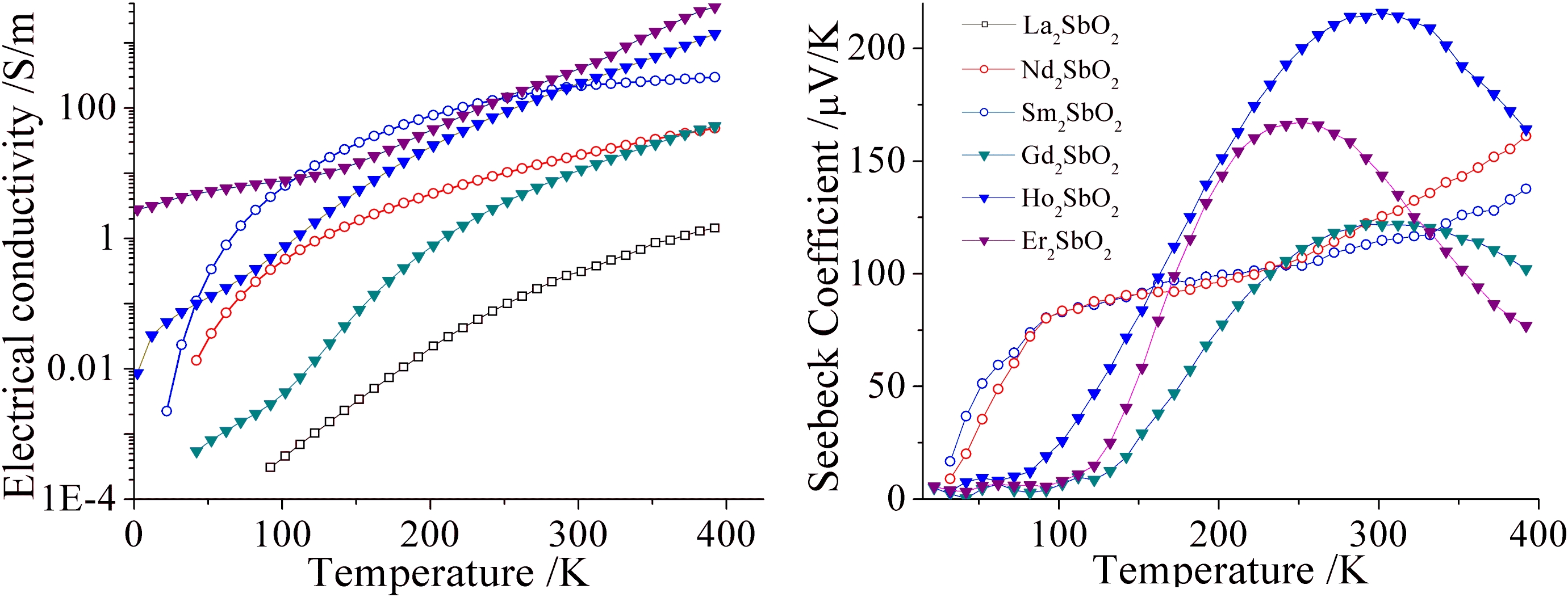
3) Tuning electron transport mechanism in RE2SbO2
The size of rare-earth elements was exploited as a chemical pressure to tune the Sb atomic disorder in the RE2SbO2 phases. As a result, RE2SbO2 materials with the same structure and charge carrier concentration exhibit a range of transport behaviors. The size of the unit cell is a governing factor determining the degree of the Sb site disorder and Sb-Sb distances, both of which determine the dominant charge carrier transport mechanism. As the result, rather than changing uniformly within the series, the transport properties of some analogues are unique and possess different activation processes. Additionally, the thermoelectric power of these compounds are improved without compromising the electrical conductivity. The RE2SbO2 compounds are, by no means, the most efficient thermoelectric materials. However, this series illustrates possibilities of by passing the classical challenges in the thermoelectric research. Although the actual physics in these systems may be more complex, experimentally, the decoupling of electrical conductivity and thermopower was indeed achieved by perturbing the charge carrier transport mechanism without modifying the carrier concentration.
Figure 4. Electrical conductivity and Seebeck coefficient for some RE2SbO2 phases