AmericanChemicalSociety.com
Reports: AC1 47575-AC1: A Greener Phase-Switch Concept for Multistep Synthesis Using Boronic Acids as Productive Tags
Dennis G. Hall, University of Alberta
There are increasing environmental pressures to improve the sustainability of reaction processes in synthetic organic chemistry. In response to these demands, new techniques and strategies are needed in order to accelerate and facilitate the synthesis and purification of organic compounds while minimizing the waste of solvents and chromatographic supports. Indeed, much of the wastes produced in organic reactions originate from the extensive use of silica gel and solvents employed in the chromatographic purification. Phase-switching strategies are very attractive as a means to avoid chromatographic purification. In phase-switch chemistry, reactions take place conveniently under homogeneous conditions, and product separation is facilitated by a liquid partition or a precipitation–filtration operation. All known phase-switching strategies require two chemically unproductive steps: attachment of the tag to the substrate, and detagging of the product at the end. The latter operation often leaves an undesired remnant (or "trace") of the phase-switch tag. We designed a new phase-switching strategy involving the boronic acid functionality as a productively convertible tag. Rather than cleaving the extraneous tag at the end of a synthetic sequence, as in other phase-switch systems, the boronic acid can be derivatized productively using the wide range of selective transformations known for this class of compounds. As hundreds of functionalized boronic acids are available commercially and can serve as potential substrates in many synthetic applications, this phase-switch system can also circumvent the tag attachment step.
In our 2009 Report, we described the development of a liquid-liquid, water-organic phase-switching system exploiting the known ability of boronic acids to form strong complexes with polyol additives at high pH. We found that sorbitol, a cheap non-toxic polyol, was polar enough to efficiently "phase-switch" hydrophobic boronic acids to a high-pH aqueous phase and be completely insoluble in organic solvents so as to avoid contamination of the organic layer. Further optimization identified sodium carbonate as a preferred, milder base. As of August 2009, the efficiency of this system was demonstrated in control experiments such as purification of boronic acids from complex mixtures of compounds, and small multistep syntheses. In the planning of a phase-switch synthetic cycle to a desired compound class, the initial substrate is selected from a broad choice of commercial boronic acids, and that functionality is preserved until the last step of productive tag removal. In order to demonstrate the utility of the productive phase-switch concept in target-oriented synthesis, we achieved and efficient racemic synthesis of ezetimibe, (Zetia), a commercial b-lactam-containing anti-hypolipidemic drug. Our synthetic sequence to ezetimibe emphasized the possibility of using the boronic acid tag as a masked hydroxyl group, and required five steps with a traditional chromatographic purification only at the last step. All of this work has been published earlier this year (S. Mothana, J.-M. Grassot, D.G. Hall, Angewandte Chemie International Edition 2010, 2883-2887). Since that time, the student working on the project, Mr. Sam Mothana, has been writing his thesis, and the postdoctoral assistant, Dr. Grassot, has left my group to take a position in France. However, some work was done by Mr. Mothana between September 2009 and February 2010, and this work was aimed at expanding the scope of reactions compatible with a boronic acid and thus amenable to our new phase-switch synthesis concept.
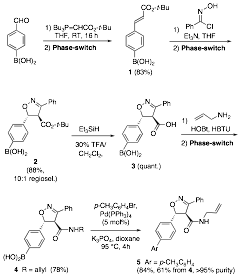
A phase-switching system employing boronic acids as phase tags must be complemented with a broad repertoire of chemical transformations tolerant of this functionality. In this regard, we have optimized a number of simple transformations of model unprotected boronic acids with aphase-switch purification. We found that alcohol oxidations, carbonyl reductions, carbodiimide-promoted esterification, carbonyl addition reactions with organometallic reagents, and even a sequence of aldehyde alkynylation and alkyne-azide cycloaddition can all be realized in good to high yields and high purify after the phase-switch purification. Since September 2009, we have optimized other reactions such as Wittig reaction and dipolar cycloadditions and applied those to a phase-switch synthesis of polysubstituted oxazolines (Scheme 1). The synthesis using the sorbitol-based liquid-liquid partition compared favorably with an earlier synthesis using the DEAM-PS resin as phase-switch conditions. The application to the Wittig reaction is particularly helpful because the phase-switching circumvents the difficult chromatographic separation of the phosphine oxide by-product associated with these olefinations.
Scheme 1. Phase-switch synthesis of polysubstituted oxazolines.
This short synthesis also further demonstrates the suitability of this concept for preparing highly functionalized molecules. In the next year, we will focus on further expanding the breadth of compatible reactions to include more redox processes such as epoxidation, alkene dihydroxylation, as well as other organometallic additions. This planned work is essential in order to demonstrate the utility of this concept and facilitate its adoption by other synthetic chemists.
Copyright © American Chemical Society