AmericanChemicalSociety.com
Reports: DNI1 48906-DNI1: Synthesis and Application of Functionalized Organocopper Species by C-Si Bond Activation of Organosilane Precursors
Zachary Ball, PhD, Rice University
The search for increasingly selective and environmentally benign methods continues to be of importance to synthetic methodology. Organosilane reagents are important in this regard because they are typically stable to a wide range of functional groups and reaction conditions, allowing selective chemical transformations in multifunctional molecules. In addition, organosilanes are generally inexpensive and produce innocuous and easily removed polysiloxane byproducts. However, the classical use of organosilanes as carbon nucleophiles for C–C bond formation is generally limited to intramolecular processes or reactions with extremely reactive electrophiles. Our work is aimed the activation and transmetalation of organosilanes with transition-metal catalysts to produce reactive organometallic intermediates from stable silane precursors. In particular, we are interested in the development of stable copper(I) fluoride complexes for the transmetallation of organosilanes, affording a new, general, and functional-group tolerant access to organocopper species. A key insight to allow development of these processes has been the use of heterocyclic carbene ligands to stabilize copper(I) species, to avoid problems of metal fluoride instability, and to control reaction selectivity.
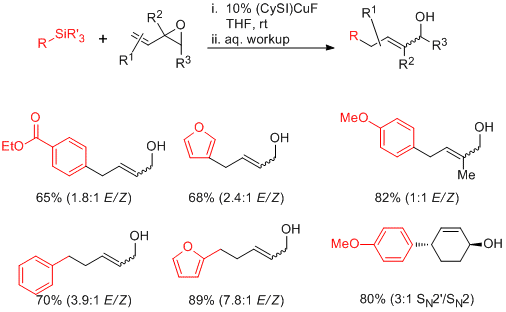
Following preliminary work from our labs which established the stoichiometric transmetalation of organosilane and copper complexes of the type LCuF (L = heterocyclic carbene), we moved toward the development of catalytic processes. Despite concerns about the ability to generate turnover in the absence of a stoichiometric fluoride activator, optimization of ligand structure resulted in the discovery that the complex (SICy)CuF is a competent catalyst for the coupling of organosilanes with vinylepoxide substrates at room temperature. Significant substitution and functional-group compatibility is tolerated, and the silane nucleophile can include (hetero)aryl and (hetero)benzylic groups.
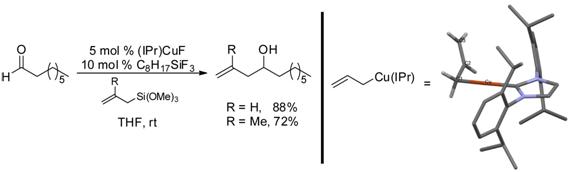
We have also examined the formation, structure and reactivity of allylcopper intermediates. Treatment of (IPr)CuF with an allylsilane results in the formation of the first synthesis of an allylcopper species, which could be characterized by NMR and crystallography. This process enabled the development of a simple protocol for catalytic aldehyde allylation.
Extension of these ideas to other substrate classes, including 1,4-addition to enone and enoate substrates, is under development and will be reported in due course. In addition to reactivity, it is found that ligand structure can be used to control reaction enantioselectivity. Functional-group selectivity can also be controlled by ligand structure, allowing the development of tandem cascade reactions triggered by activation of C–Si bonds.
Copyright © American Chemical Society