www.acsprf.org
Reports: AC947421-AC9: Stability Behavior of Clay/Nanoparticle Suspensions
John Y. Walz , Virginia Polytechnic Institute and State University
Overview of Project
The goal of this project is to understand the formation mechanisms and properties of freeze-cast and sintered composites produced from binary mixtures of silica nanoparticles and kaolinite clay platelets. The composites display a porous structure arising from the formation and propagation of ice crystals during freezing (termed freeze casting). The addition of kaolinite clay particles to the dispersion alters significantly the size and shape of the pores, as well as the mechanical strength of the composite.
Our primary focus this past year was on understanding the properties of sintered ceramics produced from these freeze-cast nanocomposites. The results of our studies are summarized below.
1. Effects of the Kaolinite Platelets on the Sintering Process
Experimental Procedure
Aqueous suspensions containing varying concentration of kaolinite and silica nanoparticles were prepared and allowed to gel. The total solids content was fixed at 18 vol%. Once gellation was complete, the samples were cooled to 238 K under vacuum for 400 min. For sintering, these freeze-cast samples were heated to either 1000 or 1250oC for 1 h.
Structure of Sintered Nanocomposites
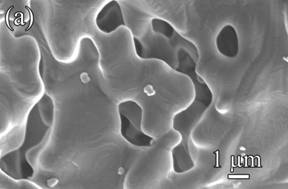
Shown in Figure 1 are scanning electron microscopy images taken on samples containing varying amounts of kaolinite before (left hand) and after (right hand) sintering to 1000oC. For samples containing only silica ( Fig. 1a), the silica nanoparticles grow to 200 nm to 600 nm grains. Individual silica nanoparticles can no longer be identified.
For the pure kaolinite sample (Fig. 1b), the boundaries between kaolinite platelets become less distinct after sintering and the sharp edges and corners become rounded. It is speculated that some sintering diffusion and inter-platelet bonding occurs.
For the 10 vol% kaolinite sample (Fig. 1c), the grain growth of the silica nanoparticles does not occur and the nano-sized silica particles are conserved. In fact, the size of the silica was actually found to decrease slightly.
Fig. 1. Microstructures of: (a) pure silica sample, (b) pure kaolinite sample, and (c) 10 vol% kaolinite-silica nanocomposite. The left and right hand side images were taken before and after sintering, respectively.
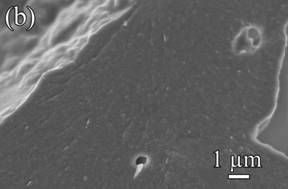
Interestingly, we found that this hindrance effect did not extend to higher temperatures. Fig. 2 displays the microstructure of samples sintered at 1250oC. Compared to samples sintered at 1000oC, both the grains and pores of the pure silica sample grow even larger. In both the pure kaolinite sample (Fig. 2(b)) and the 10 vol% kaolinite sample (Fig. 2(c)), the individual kaolinite platelets can no longer be observed, indicating that kaolinite-kaolinite and/or kaolinite-silica diffusion is promoted at elevated temperatures. The lack of discernible boundaries suggests that the two components have merged to form a new phase.
Fig. 2. Microstructures of: (a) pure silica sample, (b) pure kaolinite sample, and (c) 10 vol% kaolinite-silica nanocomposite sintered at 1250ºC for 1 h.
Proposed Explanation of Results
X-ray diffraction and differential scanning calorimetry studies, in conjunction with the SEM images show above, led us to the following conclusions.
- Pure silica nanoparticles experience grain and pore growth when sintered at 1000ºC, transforming from an amorphous structure into a tetragonal crystalline structure at approximately 850ºC. In the presence of sufficient concentrations of kaolinite platelets (roughly 4 vol% kaolinite), however, these phase transformation is almost completely inhibited. Nonetheless, reductions in the size of the nanoparticles after sintering suggest that diffusion between the nanoparticles and kaolinite platelets occurs.
- Upon increasing the sintering temperature to at least 1200oC, boundaries between the two species can no longer be discerned in the SEM images. In addition, new features are observed in the XRD pattern of the composite material that cannot be seen in the patterns of the silica-only or kaolinite-only samples sintered. This suggests that a new phase transformation has occurred at this higher temperature that incorporates both the silica and kaolinite materials.
2. Effects of the Kaolinite Platelets on the Strength of the Sintered Composites
Experimental Procedure
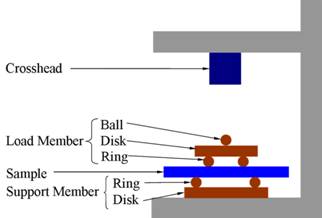
Freeze-cast nanocomposites were prepared as described above. Freeze-cast samples were sintered at either 1000oC or 1200oC for 1 h. The equibiaxial flexural strength of the freeze-cast composites was measured by a strength test apparatus with a 1 kN load cell. A schematic illustration of the instrument is shown in Fig. 3.
Fig. 3. Schematic illustration of the equibiaxial flexural strength test apparatus.
Results of Strength Measurements
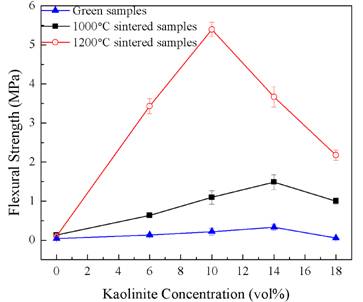
The graph shown below in Fig. 4 shows the development of the strength of these samples. As seen, sintering increases the strength for all of the samples, regardless of the kaolinite content. In addition, for all samples except the silica-only sample, increasing the sintering temperature increases the strength.
In addition, samples containing a binary mixture of kaolinite and silica are always stronger than those containing only silica or only kaolinite. For the green samples, the maximum strength is approximately 8 times higher than that of the pure silica. Our hypothesis is that the silica nanoparticles bond the structure together, while the large kaolinite platelets distribute any applied stress over a larger area. In the 1000oC sintered sample, the maximum strength is approximately 11 times greater than that of the silica-only sample.
Upon increasing the sintering temperature to 1200oC, the flexural strength of the material also increases substantially. Specifically, the strength of the 10 vol% sample is now approximately 52 times greater than the silica-only sample, and 3.6 times greater than the 14 vol% sample sintered at 1000oC.
Fig. 4. Results of equibiaxial flexural strength measurements for the freeze-cast samples as a function of kaolinite content.
Proposed Explanation of Results
It is our hypothesis that the ability of the kaolinite to increase the strength of the composites arises from two factors. First, the relatively large size and inherent strength of the platelets allows for the distribution of any applied load over a larger volume of the sample. Second, the kaolinite is able to form a new phase with the silica upon sintering at 1200oC that provides additional enhancement of strength.