Reports: AC4
48217-AC4 Development of New Pathways for the Oxidative Transformation of Alkynes into Highly Reactive Carbonyl Compounds
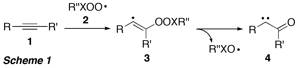
The development of novel oxidation procedures that use the most abundant (and cheapest) oxidant, molecular oxygen, under non-toxic conditions is a major goal in synthetic chemistry. In this project we have explored, whether peroxyl radicals R''XOO (2, X = S, C) can be used to transform alkynes into carbonyl compounds. Addition of R''XOO to the alkyne triple bonds in 1 should give vinyl radical 3 (Scheme 1), which, according to preliminary calculations, should undergo homolytic gamma-fragmentation of the weak peroxyl bond in 3 to form alpha-oxo carbene 4, potentially via an intermediate oxirene (not shown).
Since R''XOO are easily accessible through reaction of the respective C- or S-centred radical R''X with oxygen, this sequence would enable generation of highly reactive and synthetically very useful alpha-oxo carbenes in a single step from simple alkynes.
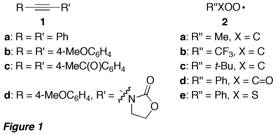
Figure 1 presents the various alkynes and peroxyl radicals used in this project. Whereas alkynes 1a-c were symmetrical diphenylacetylenes with different electron density at the pi system, alkyne 1d was expected to give an alpha-oxo carbene, which is stabilized through capto-dative effects.
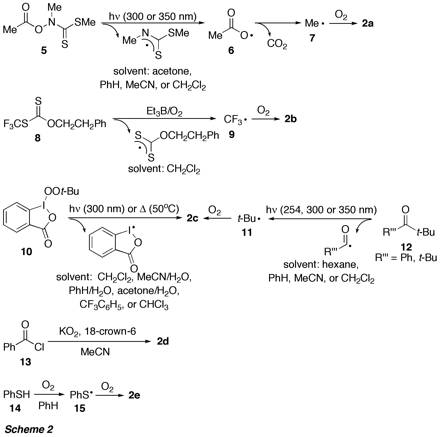
Generation of peroxyl radicals 2a-e was performed in the presence of the alkyne by both photochemical and thermal methods (Scheme 2).
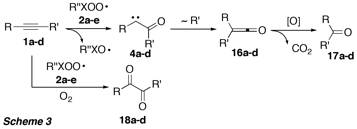
It was anticipated that intermediately formed alpha-oxo carbenes 4a-d should undergo Wolff-Rearrangement, and the resulting ketenes 16a-d should be further oxidized by the peroxyl radicals to give the corresponding ketones 17a-d after decarboxylation (Scheme 3). Thus, formation of the latter would provide indication for the success of the reaction sequence.
Interestingly, only in the reaction of diphenylacetylene (1a) with peroxyl radical 2c formation of benzophenone (17a) was observed, however with only very low yield (<10%). The rate of the addition of the electrophilic peroxyl radicals was strongly influenced by the electron density at the alkyne pi system, and with the electron-deficient alkyne 1c no reaction occurred, whereas reaction with the more electron-rich alkyne 1b was faster than with 1a. Quite unexpected, the reactions of 2c with alkynes 1a,b,d resulted in formation of the corresponding 1,2-diketones 18a,b,d. This discovery is highly relevant from a synthetic point of view, since this reaction may be considered as a very mild alternative to the existing oxidation procedures of alkynes to 1,2-diketones, which generally require harsh conditions and toxic heavy metal compounds. Most importantly, when the peroxyl radicals are produced by reaction of C- or S-centred radicals with oxygen, this methodology is can be regarded as an environmentally benign, radical mediated metal-free activation of oxygen.
Given the potential synthetic relevance and novelty of this transformation, we focussed our research towards the optimisation of reaction conditions and exploration of the mechanism of diketone formation.
a) The Role of Oxygen
It appeared that presence of oxygen was crucial for the transformation 1 -> 18 by peroxyl radicals. For example, when alkyne 1a was reacted with t-BuOO (2c), which was generated by thermal decomposition of the hypervalent iodocompound 10 in the absence of oxygen (see Scheme 2), diketone 18a was not formed and no reaction occurred.
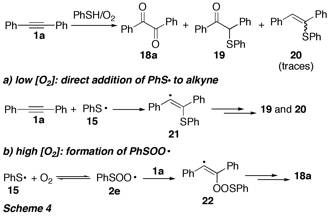
In the case of peroxyl radicals derived from thiyl radicals, PhSOO (2e), we could study the influence of oxygen in detail, since the reaction pathway strongly depended on the concentration of oxygen (Scheme 4).
Thus, when the reaction of thiyl radicals 15, which were generated by autoxidation of PhSH (Scheme 2), with alkyne 1a was performed at low [O2], direct radical addition to the alkyne occurred, resulting in formation of the thioethers 19 and 20. When [O2] was increased, the yield of diketone 18a also increased (to up to 76%), at the expense of 19 and 20. Under these conditions, the thiyl radicals 15 were trapped by oxygen to give peroxyl radicals 2e, which undergo addition to the alkyne triple bond. Computational studies using Density Functional Methods (e.g. BHandHLYP) revealed that the direct addition of 15 to alkyne 1a and the trapping of 15 by oxygen have practically identical activation barriers and reaction enthalpies and are strongly competing pathways, which could be influenced by [O2]. To our knowledge, the sequence shown in Scheme 4 is the first example for a synthetic application of thiylperoxyl radicals.
b) Mechanism of Diketone Formation
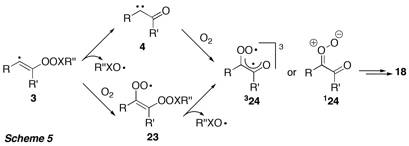
Scheme 5 outlines the suggested mechanism for the transformation of alkynes to 1,2-diketones by peroxyl radicals and oxygen.
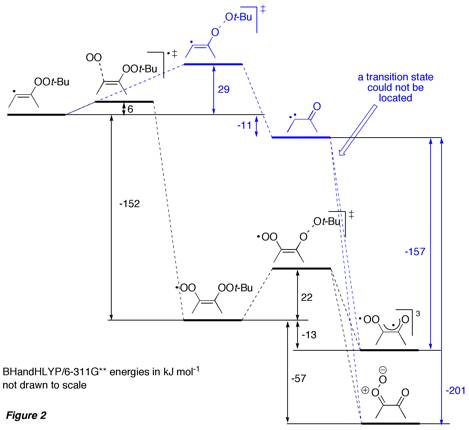
The alpha-oxo carbene 4 obtained by gamma-fragmentation in vinyl radical 3 could be trapped by oxygen to give the triplet biradical compound 324 and/or the Crigee-type singlet carbonyl oxide 124. The latter have been proposed in the literature as intermediate in the oxidation of alkynes by ozone leading to 1,2-diketones. Alternatively, vinyl radical 3 could first be trapped by oxygen to give peroxyl radical 23, followed by fragmentation of the peroxyl O–O bond. We have performed DFT computations to study the potential energy surface of the slightly simplified reaction system involving 2-butyne and peroxyl radical 2c (Figure 2).
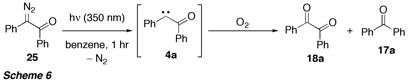
According to the calculations, trapping of the initially formed vinyl radical by oxygen appears to be more favourable than gamma-fragmentation of the O–O bond to form the carbene, which is subsequently trapped by oxygen (so far no attempts were made to study the influence of the carbene multiplicity on the reaction outcome). The prediction that under our experimental conditions a free carbene is not formed, is supported by the observation that alpha-oxo carbene 4a, which was generated by photochemical decomposition of the diazoketone 25 in the presence of oxygen always undergoes Wolff-Rearrangement, followed by oxidation/decarboxylation to benzophenone (17a). However, oxidation to diketone 18a was also observed under these conditions (Scheme 6).
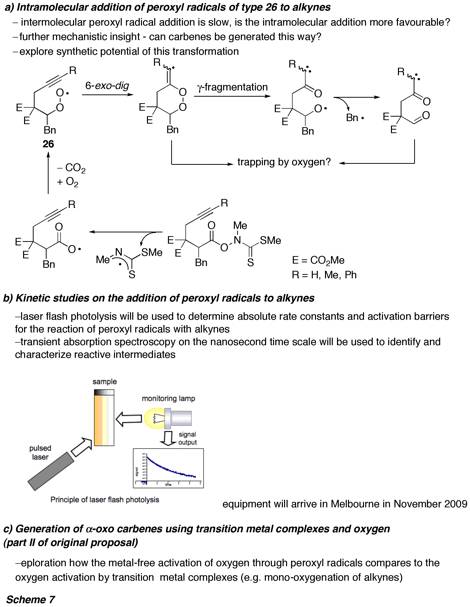
In the future, we will further explore this novel metal-free activation of oxygen for the oxidative transformation of pi systems into carbonyl compounds. The PhD student working on this project has already presented the results on various national and international conferences, and a manuscript is in preparation. This project forms the foundation of a major new research direction that is focussed around kinetic and mechanistic studies using Laser Flash Photolysis. A selection of currently ongoing and future projects is shown in Scheme 7.